Category Archives: Peptide
A Simple Protocol to Refold Peptides or Small Proteins
Understanding the process of peptide folding is a critical first step toward understanding protein folding. Depending on the temperature and solvent conditions, peptides are highly flexible and can adopt a variety of conformations in solution. Many unfolded peptides could spontaneously refold in vitro to form a native protein with full biological activity in the absence of other factors. Peptide fragments of proteins often have intrinsic propensities for the formation of their native conformations.
Peptide Folding
Proteins are the workhorses inside living cells. The interactions among proteins are critical for various important biological processes. Almost about 15-40% of the protein-protein interactions are peptide-mediated. A short stretch of amino acid residues from one protein partner contributes most to its binding to the other protein structure. These short linear interacting motifs can be found embedded inside disordered regions of intrinsically disordered proteins, or appear as flexible linkers connecting function regions and as flexible loops to rigid fragments and domains.
The primary sequence contains all the information to define the three-dimensional structure of a protein and its biological functions. The mutation or deletion of any amino acid may have a big impact on folding and stability. It takes nanoseconds (ns) for the peptide to form an intermolecular contact. The timescale of loop closing is 10 nanoseconds (ns). The formation of alpha-helical peptides is 200 ns, beta hairpins, and mini-proteins in 1–10 ms timescale. Many studies had a very good agreement between measured and calculated folding rates. Many factors such as temperature, pH, molecular chaperones, salts, and denaturants may affect a peptide in reaching its native state.
So it is critical to minimize factors that affect protein refolding. A successful folding should have inadequate denaturant concentrations to destabilize the native state of a peptide or protein. GuHCL can be used in order to disrupt the hydrophobic interactions within the tertiary structure.
- The peptide was solubilized in resuspension buffer (50 mM Tris, pH 8, 6 M GuHCl (Sigma, G4505), 10 mM DTT, 2mM EDTA) by vortexing.
- Use enough resuspension buffer such that the final peptide concentration is 0.2mg/ml.
- The resuspended peptide was then diluted 50% in dialysis buffer #1 (50 mM Tris, pH 8, 2 M GuHCl, 2mM EDTA) resulting in a 4 M GuHCl-containing solution.
- The peptide solution was then dialyzed overnight at 4°C in snakeskin dialysis tubing (Pierce) against 2 L of buffer #1.
- The following day the dialysis buffer was changed to 2 L of dialysis buffer #2 (50 mM Tris, pH 8, 1 M GuHCl, 0.4 M Arginine (Sigma, A5006), 3 mM Reduced Glutathione, 0.9 mM Oxidized Glutathione, 2mM EDTA) for overnight dialysis at 4°C.
- The following day the dialysis buffer was diluted 50% with water and dialysis continued overnight.
- Any insoluble material was centrifuged (18000×g at 2–8°C for 20 minutes) and the remaining peptide solution dialyzed overnight at 4°C against 1 L of dialysis buffer #3 (50 mM Tris, pH 8, 250 mM NaCl, 0.1 M Arginine, 3 mM Reduced Glutathione, 0.9 mM Oxidized Glutathione, 2mM EDTA) to remove the remaining GuHCl.
- The final dialyzed protein solution was clarified by centrifugation (18000×g at 2–8°C for 20 minutes) and the supernatant was separated by RP-HPLC.
References:
- Current Opinion in Structural Biology 2003, 13:168–174
- Reversible Peptide Folding in Solution by Molecular Dynamics Simulation, J. Mol. Biol. (1998) 280, 925-932
- A Simplified Method for the Efficient Refolding and Purification of Recombinant Human GM-CSF.
Protein Refolding for Western Blotting
For refolding, proteins in the SDS-Polyacrylamide gels were incubated in transfer buffer I (0.01% Triton X-100, 48 mM Tris, 39 mM Glycine, 20%methanol, pH 9.2) twice for 15 min, and then transfer buffer II (48 mM Tris, 39 mM Glycine, 20%methanol, pH9.2) twice for 15 min, then transferred onto Immobilon-P membranes (Millipore) in transfer buffer II and processed by standard procedures for Western blots.
Reference: Zhou J, Blissard GW. Mapping the conformational epitope of a neutralizing antibody (AcV1) directed against the AcMNPV GP64 protein. Virology. 2006 Sep 1;352(2):427-37. doi: 10.1016/j.virol.2006.04.041. Epub 2006 Jun 14. PMID: 16777166; PMCID: PMC3767133.
Our Services:
Custom Peptide Synthesis Services
Other Posts: To Make Simpler and Better Biosensors LifeTein Peptides Used for Pulldown Assay Peptide Applications
LifeTein Peptides Used for Pulldown Assay
Pulldown Experiments With LiteTein Peptides
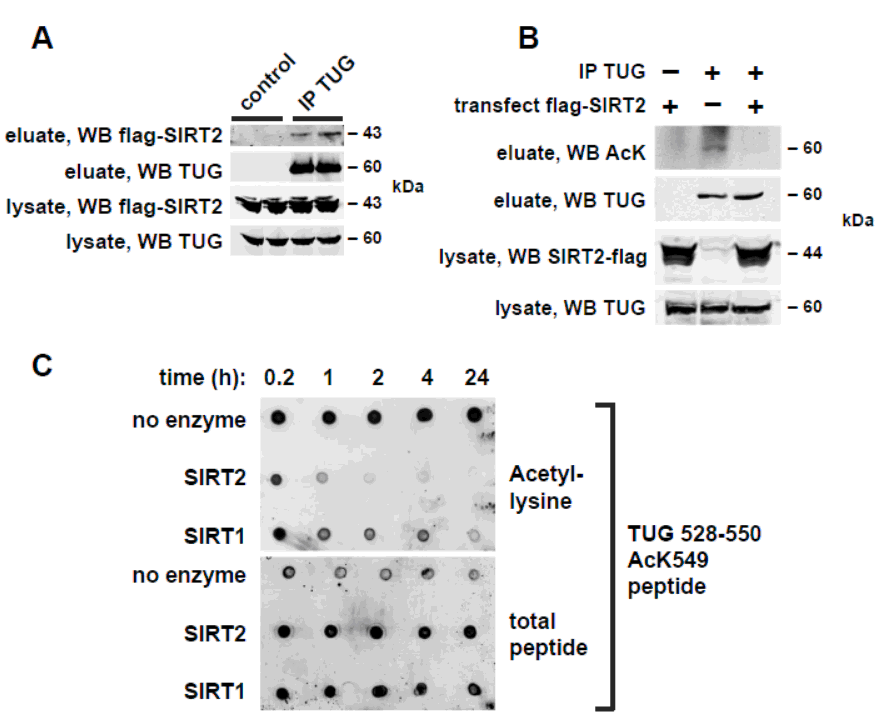
These peptides were bound to streptavidin-agarose beads and used for pulldown experiments using cell lysates. Peptides, streptavidin agarose beads, and cell lysates were permitted to bind for overnight at 4 °C, then beads were pelleted, washed five times using spin columns, and proteins were eluted in sample buffer and analyzed by SDS-PAGE and immunoblotting. All synthetic peptides had an N-terminal biotin. After incubation, peptides were spotted on nitrocellulose membranes and immunoblotted to detect acetylated Lysine (AcK) or total biotinylated peptide. Data is shown below. Reference: Acetylation of TUG Protein Promotes the Accumulation of GLUT4 Glucose Transporters in an Insulin-Responsive Intracellular Compartment, The Journal of Biological Chemistry, January 5, 2015, doi: 10.1074/jbc.M114.603977 jbc.M114.603977. Our Services: COVID-19 Services & Products Custom Antibody Services Rush Peptide Synthesis Peptide Nucleic Acids (PNAs) Custom Peptide Synthesis Services Gene Synthesis Service Custom Chemical Synthesis Other Posts: Amino acid composition of cell-penetrating peptides (CPPs) To Make Simpler and Better Biosensors A Simple Protocol to Refold Peptides or Small Proteins Peptide ApplicationsSynthetic Peptides Used for indirect ELISA
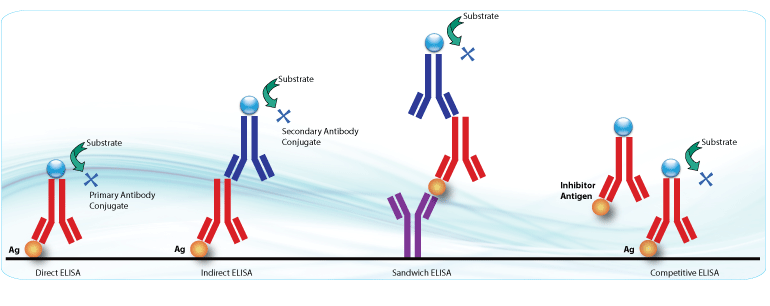
Peptides can be used for ELISA assay. A peptide-based indirect ELISA was used to screen a population of 40 Multiple sclerosis patients and 39 healthy controls. The encephalitogenic myelin oligodendrocyte glycoprotein (MOG)35–55 synthetic peptides were synthesized by LifeTein.
LifeTein’s Synthetic Peptides for ELISA
All MOG peptides or Mycobacterium avium subspecies paratuberculosis (MAP) peptides were synthesized at >90% purity by LifeTein to make sure the ELISA results are clean and consistent. The plates were coated with peptides. After overnight incubation, the plate was blocked, rinsed, and late react with antibodies according to the protocol. In silico analysis identified two peptides belonging to MAP and BCG, which share sequence homology with MOG(35–55). The peptide-based indirect ELISA data showed that sharing of highly conserved linear amino acidic sequences is necessary to elicit antibody-mediated cross-reactivity.
These findings concluded that the presence of MOG (35–55)-specific antibodies in multiple sclerosis pathogenesis. This can be used as a diagnostic biomarker in multiple sclerosis.
[PDF] Evaluation of the humoral response against mycobacterial peptides, homologous to MOG35–55, in multiple sclerosis patients
MG Marrosu, LA Sechi – 2014
… All peptides were synthesized at N90% purity commercially (LifeTein, South
Plainfield, NJ 07080 US). Purified peptides were prepared as [10 mM] stock solutions,
and were stored in single- use aliquots at −80 °C. 2.3. ELISA …
Our Services:
Custom Peptide Synthesis Services
Other Posts:
LifeTein Peptide Cited in Cell
Phospho-specific antibodies by LifeTein published in Nature
Synthesis of multiple antigenic peptides: strategies and limitations
The Structural Basis of Peptide-Protein Binding Strategies
LifeTein Peptide Cited in Cell
Peptide by LifeTein Cited in Cell
Overlapping peptides from LifeTein were used to map the region of Fragment 3 by epitope mapping of anti-Fzd2 antibody. This anti-Fzd2 antibody was found to reduce tumor growth. Wnt signaling plays a critical role in colorectal cancer. Researchers found that Wnt receptor Frizzled2 (Fzd2) and its ligands Wnt5a/b are elevated in metastatic liver, lung, colon, and breast cancer cell lines. Their high level expression correlates with markers of epithelial-mesenchymal transition (EMT). By epitope mapping using synthetic peptides from LifeTein, the researchers mapped the epitope to a specific region. The antibody to Fzd2 was found to reduce cell migration and invasion. Targeting this pathway may provide a cure for patients with tumors expressing high amount of Fzd2 and Wnt5a/b. We have developed an antibody to Fzd2 that reduces cell migration and invasion and inhibits tumor growth and metastasis in xenografts. We propose that targeting this pathway could provide benefit for patients with tumors expressing high levels of Fzd2 and Wnt5a/b.- Cell, Volume 159, Issue 4, 6 November 2014, Pages 844–856, DOI: 10.1016/j.cell.2014.10.032 A Noncanonical Frizzled2 Pathway Regulates Epithelial-Mesenchymal Transition and Metastasis
Phospho-specific antibodies by LifeTein published in Nature
Jia Shen. et al. EGFR modulates microRNA maturation in response to hypoxia through phosphorylation of AGO2. Nature 497, 383–387 (16 May 2013), doi:10.1038/nature12080 LifeTein helped designed and synthesized a series of phosphorylated peptides. Then the peptides were used for phospho-specific antibody productions. The phospo-specific antibodies by LifeTein were confirmed to react with the epidermal growth factor receptor (EGFR). The Hung’s lab showed that AGO2-Y393 phosphorylation mediates EGFR-enhanced cell survival and invasiveness under hypoxia. These findings suggest that modulation of miRNA biogenesis is important for stress response in tumour cells. … The following peptides were chemically synthesized for antibody production in mice (Lifetein Conc.), Elisa verification (LifeteinConc.) and peptide competition assay in immunohistochemistry (IHC)… Supplementary information
Phospho-Specific Antibodies by LifeTein
Our Services:
Custom Peptide Synthesis Services
Other Posts:
LifeTein Peptide Cited in Cell
Synthesis of multiple antigenic peptides: strategies and limitations
The Structural Basis of Peptide-Protein Binding Strategies
Synthetic Peptides Used for indirect ELISA
Synthesis of multiple antigenic peptides: strategies and limitations
Synthesis of Multiple Antigenic Peptides
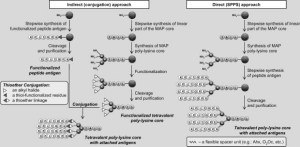
http://www.ncbi.nlm.nih.gov/pubmed/21391284 SPPS is the preferred method by LifeTein. The synthesis approach requires a branched poly-lysine core. Each branch is elongated into the corresponding epitope by stepwise SPPS. The disadvantage of this approach is that the synthetic errors could happened and cause microheterogeneity in the final materials. However the cost is lower and less time-consuming than the indirect approach. For very long linear peptides, it is more advantageous to use the SPPS method. The MAP synthesis may not always meet with success. The solubility of the peptide epitope can also become an issue and is difficult to predict for long epitopes. It is recommended to carefully design and analyze the linear epitope before MAP synthesis. Studies showed that synthesis with Ahx linker in the lysine core had better isolated yield. It is possible that the flexibilizing effect of Ahx helps in keeping peptide chains properly solvated during synthesis, preventing aggregation and hence increasing the amount of viable growing peptide sequences. Our Services: COVID-19 Services & Products Custom Antibody Services Rush Peptide Synthesis Peptide Nucleic Acids (PNAs) Custom Peptide Synthesis Services Gene Synthesis Service Custom Chemical Synthesis Other Posts: LifeTein Peptide Cited in Cell The Structural Basis of Peptide-Protein Binding Strategies Synthetic Peptides Used for indirect ELISA Phospho-specific antibodies by LifeTein published in NatureThe Structural Basis of Peptide-Protein Binding Strategies
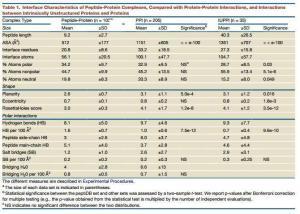
Peptide-Protein Binding
Highlights
- After binding its partners, most peptides do not introduce any conformational changes
- The interfaces of peptide-protein have more hydrogen bonds
- The peptide hotspots are important for the binding
- Peptides prefer to bind in the largest pockets on the protein surface
LifeTein is pleased to offer a free, comprehensive web-based peptide analysis tool. This tool will allow your research team to overcome common difficulties inherent in protein analysis and peptide antigen design.
Our Services:
Custom Peptide Synthesis Services
Other Posts:
LifeTein Peptide Cited in Cell Phospho-specific antibodies by LifeTein published in Nature Synthesis of multiple antigenic peptides: strategies and limitations Synthetic Peptides Used for indirect ELISA
D-amino acid peptides to resist common proteases
About D-Amino Acid Peptides
The design of all-D-peptides has been applied to increase bioactive peptides’ resistance to endogenous enzymes, as well as their bioavailability. Retro-inverso peptides are obtained by replacing the standard L-amino acid residues with the corresponding D-amino acids and reversing the direction of the peptide backbone. Therefore, the original spatial orientation and the chirality of the side chains is unchanged. This results in a non-complementary side chain topochemistry between the analog and the parental L-peptide. The significantly improved biostability of D-peptides usually leads to longer in vivo circulation half-time, making the D-peptide based drug delivery system more attractive and efficient than their L-peptide counterparts. Success has been achieved immunologically in using retro-inverso peptides toward antigenic mimicry of their parent l-peptides. It was found that the retro-all-d-peptide isomer of p53(15–29), like its parent l-peptide, adopted a right-handed helical conformation in the complex. However, in some cases, the retro-inverso isomers are significantly inferior to their parent l-peptides. The low cellular uptake of D-peptides remain an unmet challenge. So the best way is to insert some key D amino acids into the peptide sequence. For example, some pharmaceutical important peptide antibiotics such as gramicidins, actinomycins, or bacitracins incorporate D-amino acids into the drug design. The assemblies of D-peptides deserve further exploration and may lead to more surprises. See more details from here: http://lifetein.com/Peptide-Synthesis-D-Amino-Acid.html Reference: http://www.pnas.org/content/102/2/413.full.pdf+html Google+ Our Services: COVID-19 Services & Products Custom Antibody Services Rush Peptide Synthesis Peptide Nucleic Acids (PNAs) Custom Peptide Synthesis Services Gene Synthesis Service Custom Chemical Synthesis Other Posts: Amino acid composition of cell-penetrating peptides (CPPs) Obesity Treatment: A New Peptide Drug Peptide Synthesis for Cell-penetration StudiesAmino acid composition of cell-penetrating peptides (CPPs)
About Cell-Penetrating Peptides
The following table shows a selection of currently known CPPs, their origins, and sequences.| Name |
Origin |
Sequence |
| Tat (48-60) |
HIV-1 protein |
GRKKRRQRRRPPQQ |
| plsl |
Igl-1 homeodomain |
RVIRVWFQNKRCKDKK |
|
STR-R8 |
Stearly-RRRRRRRR-amide (Stearyl = CH3(CH2)16CO-) |
|
|
STR-(RxR)4 |
Stearly-(RxR)4-amide (X=6-aminohexanoic acid) |
|