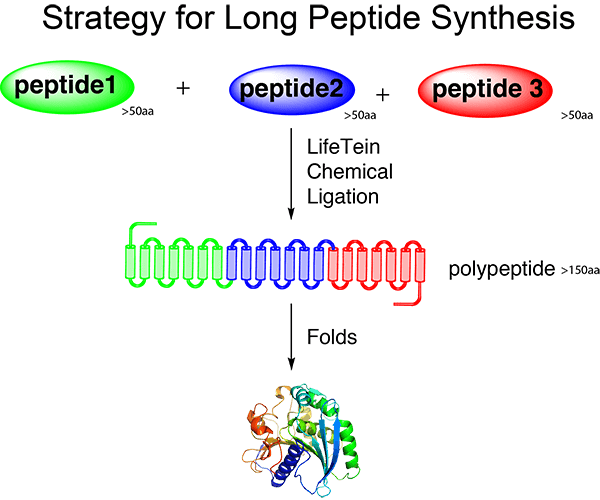
LifeTein provides long peptide synthesis services for research projects requiring peptides over 100 amino acids. Using solid-phase peptide synthesis (SPPS) together with AI-assisted peptide design and manufacturing, we support challenging long peptide projects for protein interaction studies, receptor binding studies, domain analysis, and other advanced applications.
| Peptide length | Long peptides exceeding 100 amino acids, including peptides up to 169 amino acids |
| Technology | SPPS together with AI-assisted peptide design and manufacturing, hybrid synthesis and ligation strategies for selected difficult sequences |
| Quality control | HPLC purification and molecular weight confirmation by Electrospray Ionization (ESI) mass spectrometry |
| Applications | Protein-receptor binding studies, domain-level analysis, difficult sequence synthesis, and custom modified peptide projects |
| Project review | Technical discussion and quotation support for sequence-dependent long peptide projects |
Tip: How to detect small peptides clearly and sensitively by Western blotting or SDS-PAGE?
Solid-phase peptide synthesis is the most common method of peptide synthesis today. In SPPS, the C-terminus of the first amino acid is coupled to an activated solid support such as polystyrene. The support acts as the C-terminal protecting group and allows rapid separation of the growing peptide product from reaction mixtures during synthesis.
Long peptides are frequently synthesized to study protein-receptor binding, domain interfaces, and functions that cannot be represented adequately by short peptide fragments alone. For many research projects, synthetic long peptides offer a direct alternative to cloning and overexpression workflows.
LifeTein routinely uses proprietary technologies to produce peptides longer than 100 amino acids. Our notable achievements include a 76aa BfUbb peptide, a 97aa peptide mB Box-97syn, a 133aa SHT peptide, and a 169aa long peptide. We provide HPLC-purified peptides with molecular weight confirmation by Electrospray Ionization (ESI) mass spectrometry. Net Peptide Content (NPC) is not tested by Amino Acid Analysis (AAA) unless requested separately.
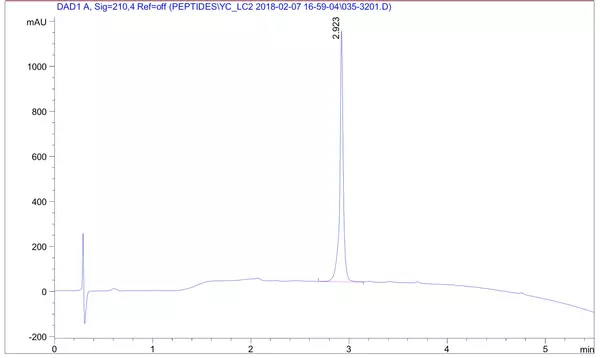
LifeTein chemically synthesized a peptide encompassing the human IRF2BPL HCD core region (a.a. 395–448), a difficult 54 amino acid sequence, with N-terminal acetylation, C-terminal amidation, and >95% purity. This is a representative example of long peptide synthesis for publication-grade biochemical research.
1. A 97AA peptide mB Box-97syn was synthesized for ease of larger-scale production and to >95% homogeneity by HPLC.
2. A 76-aa BfUbb peptide corresponding to the mature molecule without the signal sequence was synthesized by LifeTein.
3. NUCLEIC ACID VECTORS AND USES THEREOF: A peptide of 133 amino acids was synthesized by LifeTein. The peptide sequence includes at least one cell-penetrating peptide (CPP) domain, one secretion signal sequence, one DNA binding domain, and other SHT protein features.
4. HYBRID PROTEINS AND USES THEREOF: Long peptides of >100 amino acids were synthesized by LifeTein for DNA binding, transfection, and transformation studies.
Need a quotation for a long peptide?
Send us your sequence, desired purity, quantity, and any modification requirements. Our team can review whether SPPS, hybrid synthesis, or another sequence-specific strategy is most suitable.
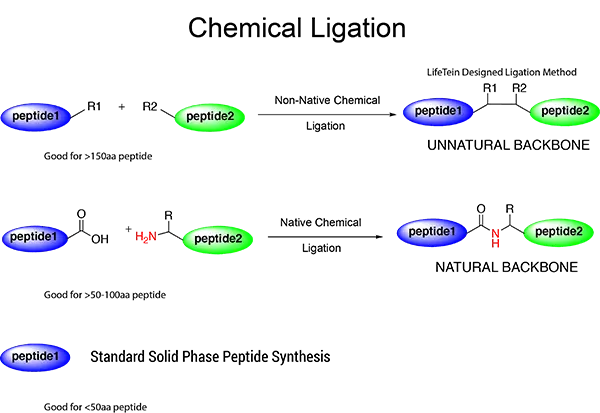
We are frequently asked to synthesize relatively large peptides sized 100–200 residues, approximately the size of a mean protein domain. We recently synthesized a 169-amino-acid peptide. This demonstrates that peptides in the 100–200 amino acid range are within the capabilities of our current PeptideSyn technology for suitable sequences. This long peptide was synthesized from intermediate fragments generated using stepwise SPPS, purified in their unprotected form, and ligated chemically with minimal protection.
A Simple Protocol to Refold Peptides or Small Proteins.
Obtaining peptides sized 100–200 amino acids by chemical synthesis can be faster and more direct than cloning and overexpressing in Escherichia coli. Chemical synthesis can also incorporate non-genetically encoded structures, such as D-amino acids, in a completely regular fashion. Synthetic peptides can avoid problems such as poor expression, cloning errors, affinity tags like FLAG or 6-His, and mis-translation of non-preferred codons in prokaryotic hosts. Artificial amino acids with isosteric side chains can be used to investigate the functional importance of specific residues.
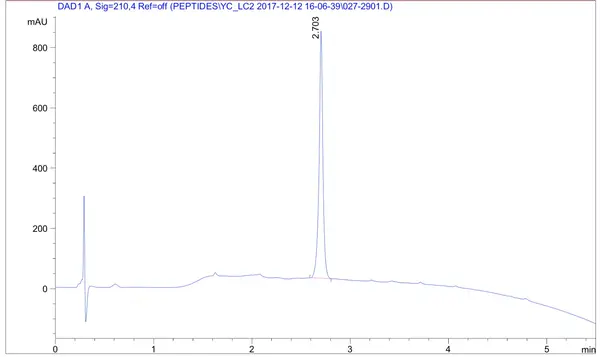
A 169-amino-acid peptide (MW 18716.41 Da, 83% purity) was synthesized successfully in 4 weeks. The success of the synthesis depends strongly on the sequence: some 150-amino-acid peptides can yield better results than peptides with only 70 residues. Usually, Boc/Bzl protection is optimal for selected long or difficult sequences and base-sensitive peptides. During deprotection and cleavage from the resin, reactive intermediates can affect vulnerable moieties such as Arg, Asp, or Glu. LifeTein’s PeptideSyn technology is designed to reduce these long-peptide synthesis challenges.
A client requested a very hydrophobic 68-amino-acid peptide with FITC modification at the N-terminus and a target purity of 85%. The peptide was synthesized successfully in 4 weeks.