Custom peptides containing non-natural amino acids, N-methyl residues, stapling building blocks, stable isotope labels, PTMs, fluorophores, and other specialized modifications.
LifeTein provides custom peptide synthesis using a broad range of special amino acids and peptide modifications. These modifications can improve peptide stability, enhance binding selectivity, support structure-function studies, enable detection, or introduce chemical handles for downstream conjugation.
Peptide stapling is a macrocyclization strategy used to constrain short peptides, often into an α-helical conformation. By covalently linking two side chains positioned on the same face of a helix, stapling can increase helicity, improve proteolytic resistance, enhance target affinity, and in some cases improve cellular uptake.
Hydrocarbon stapled peptides are commonly prepared using ring-closing olefin metathesis between two alkene-containing non-natural amino acids. Common building blocks include S5, R8, and related α,α-disubstituted amino acids.


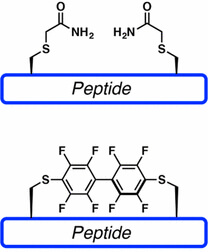
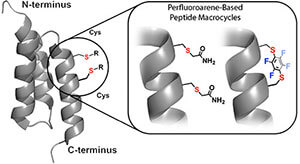
LifeTein also supports selected cysteine-reactive macrocyclization strategies, including perfluoroarene-based peptide stapling. This method uses cysteine-selective chemistry to form rigid perfluoroaromatic linkers. These staples introduce lipophilic and conformationally restrictive elements that may improve binding, stability, and cell permeability depending on the peptide sequence.
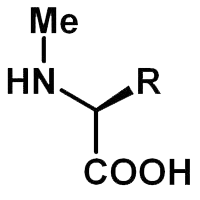
N-methyl amino acids are useful for increasing peptide rigidity, modifying hydrogen-bonding patterns, improving protease resistance, and altering membrane permeability. N-methylation is commonly used in peptide drug discovery, structure-activity relationship studies, and peptidomimetic design.
Because N-methyl residues are more sterically demanding than standard amino acids, synthesis often requires adjusted coupling conditions and careful sequence planning. Side-chain protecting groups and resin strategy may need to be optimized for difficult sequences.
Special amino acids can be used to modify peptide charge, hydrophobicity, conformational flexibility, enzymatic stability, receptor binding, and biological activity. They are especially useful in peptide optimization, epitope mapping, SAR studies, inhibitor design, and biomarker assay development.
Cysteine-containing peptides often require special planning because free thiols can oxidize, form disulfides, dimerize, or react with maleimide and other thiol-reactive groups. Protected cysteine derivatives and thiol-specific labels can help control peptide behavior during synthesis and downstream conjugation.
Cys(Acm) is commonly used when disulfide formation must be blocked or directed selectively. Free cysteine residues can also serve as handles for dye labeling, PEGylation, biotinylation, protein conjugation, or peptide-drug conjugation.
LifeTein offers selected reactive peptide modifications for protease inhibitor and activity-based probe applications.
These modifications are sequence- and application-dependent. Please provide the target enzyme, intended assay, and desired peptide format when requesting a quote.
LifeTein offers stable isotope-labeled peptides containing amino acids enriched with 13C, 15N, or other stable isotopes. These labeled peptides are commonly used as internal standards for mass spectrometry, quantitative proteomics, peptide mapping, biomarker validation, and post-translational modification studies.
Stable isotope-labeled peptides retain chemical and biological properties similar to the native peptide while allowing precise detection and quantification by MS-based workflows.
Special amino acid peptides can also be combined with fluorescent dyes, biotin, quenchers, or reporter groups for imaging, binding assays, FRET assays, enzymatic assays, and peptide tracking.
For a more complete dye-focused page, see: fluorescent peptide labeling services.
LifeTein supports selected peptide post-translational modifications for immunology, cancer biology, epigenetics, signaling, and protein interaction studies.
Please send your peptide sequence, required modifications, modification positions, desired purity, quantity, and intended application. For stapled peptides, please indicate the desired staple positions or provide the parent linear sequence for design review.