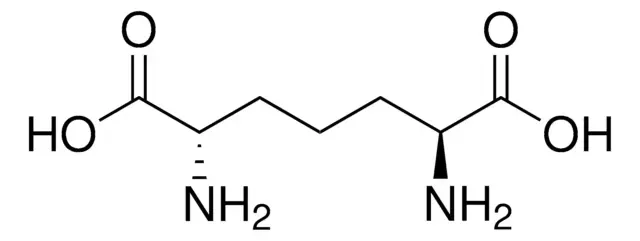
2,6-Diaminopimelic acid (DAP) is a unique non-proteinogenic diamino acid that occupies a unique position at the intersection of bacterial physiology, antibiotic development, and synthetic peptide chemistry. Structurally characterized as an α,α′-diamino dicarboxylic acid with the molecular formula C7H14N2O4, DAP is distinguished by the presence of two chiral centers and two amino groups flanking a seven-carbon backbone. This unusual amino acid is an essential constituent of bacterial peptidoglycan and serves as the biosynthetic precursor to L-lysine in both bacteria and higher plants. Importantly, mammals lack this entire metabolic pathway and require lysine as a dietary essential amino acid, making DAP and its processing enzymes attractive targets for antibiotic development with minimal mammalian toxicity. Beyond its native biological roles, DAP has emerged as a valuable building block in synthetic peptide chemistry, enabling the creation of structurally constrained peptides with potential immunostimulant, antitumor, and sleep-inducing activities.
Key Takeaways
- 2,6-Diaminopimelic acid is a symmetrical diamino dicarboxylic acid with the molecular formula C7H14N2O4 and CAS number 583-93-7, existing in three stereoisomeric forms: LL-, DD-, and meso-DAP.
- The meso-DAP isomer is an essential cross-linking component of peptidoglycan in Gram-negative bacterial cell walls, while Gram-positive bacteria typically use L-lysine for this function.
- DAP is the biosynthetic precursor to L-lysine in bacteria and plants via the diaminopimelate pathway, a route completely absent in mammals.
- Analogues of DAP, including fluorinated, phosphonate, and heterocyclic derivatives, have demonstrated significant antibacterial activity by inhibiting key enzymes in the DAP/lysine biosynthesis pathway.
- In peptide synthesis, DAP enables the construction of unusual peptides with C-termini at both ends, as well as complex structures incorporating diketopiperazine rings, which exhibit diverse biological activities.
- Custom synthesis of DAP-containing peptides for research applications is available through specialized providers like LifeTein, enabling studies of bacterial enzymes, immunomodulatory compounds, and potential therapeutic agents.
Chemical Fundamentals of 2,6-Diaminopimelic Acid
Structural Characteristics and Stereochemistry of DAP
2,6-Diaminopimelic acid is defined by its symmetrical structure: a seven-carbon dicarboxylic acid backbone with amino groups at both the C2 and C6 positions. The IUPAC name, 2,6-diaminoheptanedioic acid, accurately describes this arrangement.
The presence of two stereogenic centers gives rise to three possible stereoisomers: the LL-isomer (both chiral centers in the L-configuration), the DD-isomer (both in the D-configuration), and the meso-isomer (one center L, one center D). This stereochemical diversity is biologically critical, as different isomers serve distinct functions in bacterial metabolism and cell wall architecture.
Natural Occurrence and Biosynthetic Context
DAP is a naturally occurring amino acid found in both bacteria and higher plants. Its discovery by Work in 1950 marked an important milestone in understanding bacterial cell wall composition. In nature, DAP is biosynthesized through the diaminopimelate pathway, which converts pyruvate and L-aspartate into L-lysine, with meso-DAP serving as the immediate precursor to lysine.
The pathway involves several enzymes that have become targets for antibiotic development, including diaminopimelate epimerase (DapF), which catalyzes the interconversion of LL-DAP and meso-DAP, and diaminopimelate decarboxylase, which converts meso-DAP to L-lysine.
Find out more about peptide synthesis here.
Biological Significance and Therapeutic Relevance of DAP
Essential Role in Bacterial Peptidoglycan
The most critical biological function of DAP lies in its contribution to bacterial cell wall integrity. In Gram-negative bacteria, the meso-DAP isomer is incorporated into the peptidoglycan layer, where it acts as a cross-linking agent between glycan strands. Specifically, meso-DAP in the pentapeptide of peptidoglycan provides the attachment site that links the inner and outer membranes to the rigid cell wall structure.
This function is absolutely essential for bacterial survival; disruption of DAP incorporation or biosynthesis leads to weakened cell walls and osmotic lysis. Importantly, because mammals lack DAP and its metabolic pathway, inhibitors targeting DAP utilization exhibit minimal mammalian toxicity, fulfilling the ideal criteria for antibiotic development.
The DAP/Lysine Biosynthetic Pathway as an Antibiotic Target
The diaminopimelate pathway represents a validated target for novel antibacterial agents . Multiple enzymes in this pathway have been structurally characterized and exploited for inhibitor design:
Research has demonstrated that certain DAP analogues achieve up to 75% growth inhibition against bacterial strains, validating this approach for antibiotic discovery.
DAP Synthetic Approaches and Peptide Applications
Stereocontrolled Synthesis of DAP-Containing Peptides
The incorporation of DAP into synthetic peptides presents both opportunities and challenges due to its two amino groups and stereochemical complexity. Researchers have developed sophisticated stereocontrolled synthetic strategies to access DAP-containing peptides with defined configurations.
One elegant approach utilizes a chiral synthon derived from L-valine, specifically, a mono-lactim ether that serves as a template for constructing the DAP framework . Alkylation with dihaloalkanes followed by elaboration yields optically active tripeptides with the general structure Val-(DAP)-Val, representing unusual peptides that are C-terminal at both ends of the chain. These compounds are valuable for studying structure-activity relationships and enzyme inhibition.
Complex Peptide Architectures
Beyond simple linear peptides, DAP enables the construction of conformationally constrained structures with diverse biological activities. Researchers have synthesized peptides incorporating a proline residue fused to a diketopiperazine ring containing the DAP skeleton. These fused ring systems resemble natural products such as brevianamides, which exhibit immunomodulatory, antitumor, and antibiotic properties.
X-ray crystallography and computational modeling of these DAP-containing peptides have revealed important structural features, including the planar conformation of diketopiperazine rings and the influence of proline residues on overall molecular geometry. Such insights guide the rational design of peptide-based therapeutics.
Biologically Active Peptide Analogues
Several classes of DAP-containing peptides have demonstrated promising biological activities:
- γ-Glutamyl-diaminopimelic acid dipeptides: Synthesized via regioselective ring-opening strategies, these compounds show potential as immunostimulating agents .
- Hydrazino-dipeptide analogues: Designed as inhibitors of DAP aminotransferase, these compounds exhibit potent, slow-binding inhibition and antimicrobial activity against both Gram-positive and Gram-negative bacteria.
- Phosphonate-containing DAP peptides: While the phosphonate analogues themselves showed weak enzyme inhibition, certain tripeptide derivatives demonstrated growth inhibition against specific bacterial strains.
- Acylated DAP peptides: Conjugation of DAP-containing peptides with lauric or palmitic acid yields compounds with immunoadjuvant activity.
Research Applications and Custom Synthesis
Tools for Studying Bacterial Enzymology
DAP and its analogues serve as indispensable tools for probing bacterial enzyme mechanisms. Researchers studying DAP epimerase, DAP dehydrogenase, and MurE rely on synthetic DAP-containing substrates and inhibitors to elucidate catalytic mechanisms and to screen for potential antibiotics. The availability of fluorinated, aziridino, and phosphonate derivatives enables detailed structure-activity relationship studies.
Availability from Commercial Sources
For research applications, high-quality DAP and custom DAP-containing peptides are available from specialized suppliers. LifeTein offers expertise in incorporating unusual amino acids like DAP into custom peptide sequences, enabling researchers to access complex structures for their specific investigations. These services include:
- Synthesis of DAP-containing peptides with defined stereochemistry
- Incorporation of DAP into cyclic peptides and constrained structures
- Production of DAP analogues for enzyme inhibition studies
- Rigorous quality control, including HPLC and mass spectrometry validation
Find out about high-speed RUSH synthesis.
Frequently Asked Questions (FAQ)
What is the difference between the three stereoisomers of DAP?
2,6-Diaminopimelic acid exists as LL-, DD-, and meso-isomers due to its two chiral centers. The LL-isomer has both amino groups in the L-configuration, the DD-isomer has both in the D-configuration, and the meso-isomer has one L and one D center. The meso-DAP isomer is the biologically relevant form incorporated into bacterial peptidoglycan and serves as the immediate precursor to L-lysine.
Why is DAP considered a target for antibiotic development?
Mammals lack the entire diaminopimelate pathway and require dietary lysine, whereas bacteria depend on this pathway to synthesize both meso-DAP (for peptidoglycan) and L-lysine (for protein synthesis). Consequently, inhibitors targeting DAP biosynthesis enzymes or DAP incorporation into peptidoglycan exhibit selective antibacterial activity with minimal mammalian toxicity.
What types of DAP analogues have shown antibacterial activity?
Several classes of DAP analogues demonstrate promising antibacterial effects, including fluorinated derivatives, aziridino-DAP (extremely potent against DAP epimerase), isoxazoline-containing analogues, phosphonate derivatives, and hydrazino-dipeptides. Some analogues achieve up to 75% growth inhibition against bacterial strains.
How is DAP incorporated into synthetic peptides?
DAP incorporation requires orthogonal protecting group strategies due to its two amino groups. Researchers typically use stereocontrolled synthesis starting from chiral synthons like mono-lactim ethers derived from valine. Specialized peptide synthesis providers such as LifeTein offer custom synthesis of DAP-containing peptides with defined stereochemistry and high purity.
Paradisi, F., Porzi, G., & Sandri, S. (2001). A new stereocontrolled synthesis of uncommon tripeptides derived from 2,6-diaminopimelic acid (2,6-DAP). Tetrahedron: Asymmetry, 12(23), 3319–3324. https://doi.org/10.1016/s0957-4166(02)00002-2
Galeazzi, R., Garavelli, M., Grandi, A., Monari, M., Porzi, G., & Sandri, S. (2003). Unusual peptides containing the 2,6-diaminopimelic acid framework: Stereocontrolled synthesis, X-ray analysis, and computational modelling. Part 2. Tetrahedron: Asymmetry, 14(17), 2639–2649. https://doi.org/10.1016/s0957-4166(03)00541-x
Chatterjee, B., Mondal, D., & Bera, S. (2021). Diaminopimelic acid and its analogues: Synthesis and biological perspective. Tetrahedron, 100, 132403. https://doi.org/10.1016/j.tet.2021.132403