Fluorescent labeling with FAM (carboxyfluorescein) has become a foundational technique for visualizing and tracking peptides in a wide range of biological systems. This bright, green fluorophore is prized for its high quantum yield, excellent photostability, and compatibility with common detection platforms like fluorescence microscopy and flow cytometry. As a standard tool for cellular imaging and molecular interaction studies, FAM-labeled peptides offer researchers high-sensitivity detection with minimal background interference. However, because FAM exists as two distinct isomers, 5-FAM and 6-FAM, understanding their subtle differences is essential for designing successful experiments.
Key Takeaways
- FAM is a bright green fluorophore (Ex/Em ~490-495 nm / 515-520 nm) widely used for peptide labeling in microscopy and flow cytometry.
- The two isomers, 5-FAM and 6-FAM, differ in their chemical linkage position to the peptide backbone, which can affect binding interactions in certain assays.
- 5-FAM is a single isomer, while 6-FAM is often preferred for custom synthesis due to its commercial availability and effective linkage chemistry.
- Conjugation is typically achieved via NHS ester chemistry targeting primary amines or maleimide chemistry for thiol-specific labeling.
- Spacers such as Ahx (aminohexanoic acid) or β-alanine are often incorporated to minimize steric interference between the bulky FAM dye and the peptide’s biological activity.
Fundamentals of FAM
What Is FAM?
FAM (carboxyfluorescein) is a fluorescein derivative that contains a carboxylic acid group, facilitating covalent conjugation to biomolecules. It exhibits excitation and emission maxima of approximately 490-495 nm and 515-520 nm, placing it in the green channel of most fluorescence detection systems. This spectral profile is compatible with 488 nm argon-ion lasers, the standard excitation source for flow cytometers and confocal microscopes, making FAM an accessible and cost-effective option for routine imaging applications.
Furthermore, LifeTein offers FAM among its extensive range of fluorescent labeling services, alongside FITC, TAMRA, cyanine dyes, and Alexa Fluor dyes, enabling researchers to select the optimal label for their specific experimental design.
Why Use FAM for Peptide Labeling?
Several key attributes make FAM exceptionally suited for peptide research:
- High brightness – provides strong signal even at low concentrations.
- Good photostability – maintains fluorescence under prolonged illumination.
- pH sensitivity – fluorescence is optimal in the range pH 7.5–8.5 but decreases below pH 7, making it ideal for physiological conditions.
- Wide compatibility – works seamlessly with fluorescence microscopes, plate readers, and flow cytometers.
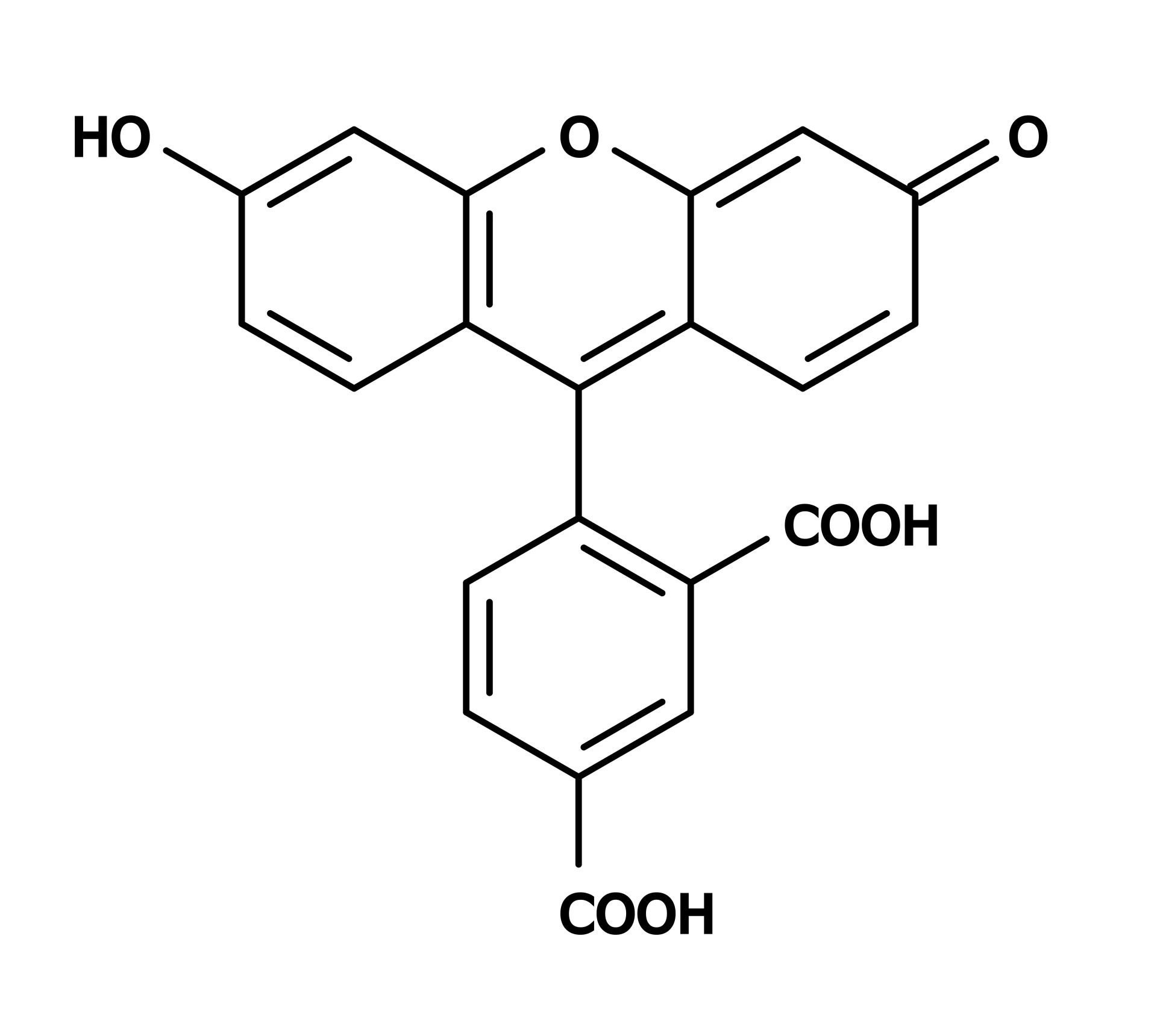
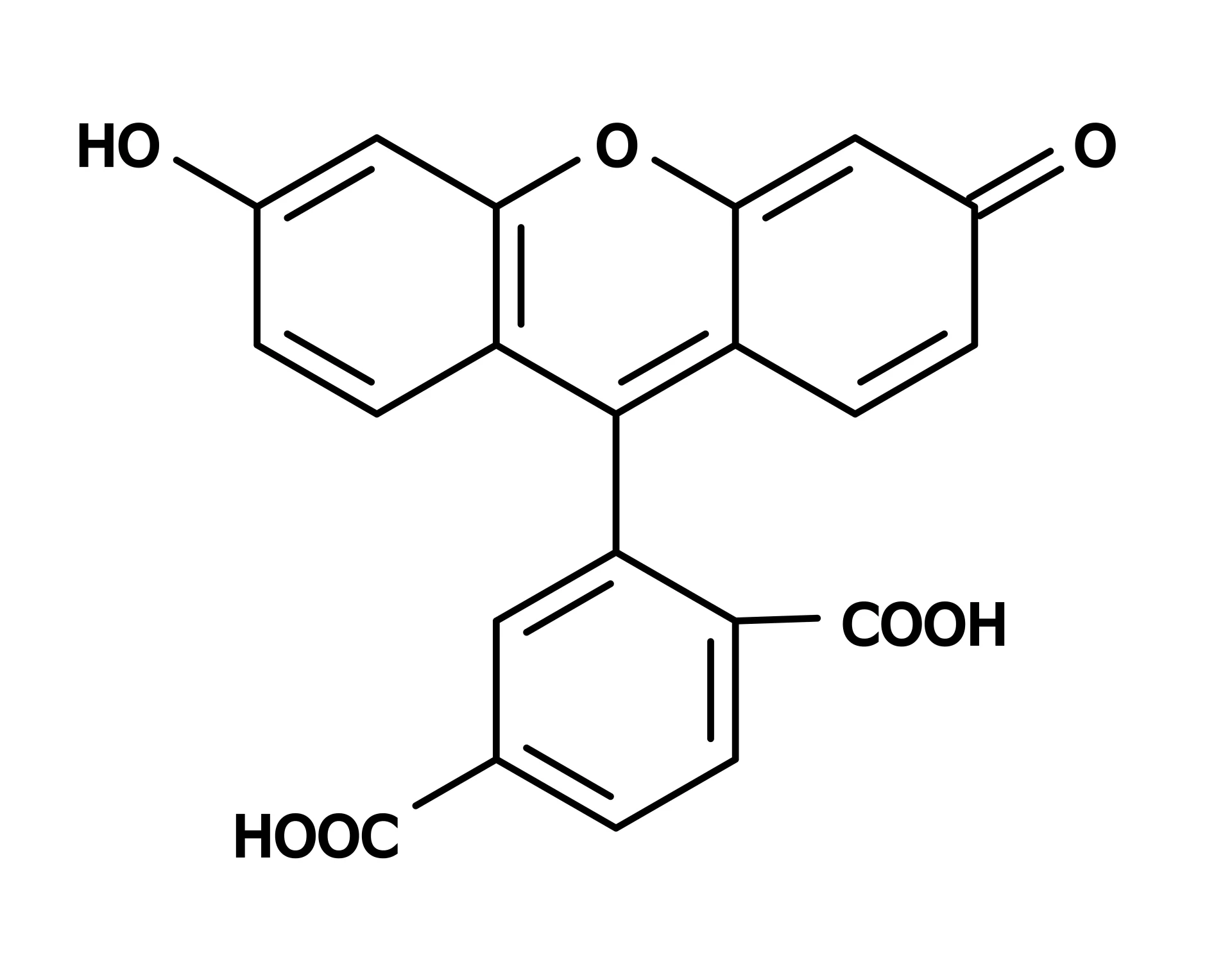
5-FAM vs. 6-FAM: A Critical Distinction for Labeling
A common point of confusion is the difference between 5-FAM and 6-FAM. Both are positional isomers of carboxyfluorescein, differing only in where the carboxylic acid group is attached to the fluorescein core (C5 vs. C6 position). This difference in linkage position means that when conjugated to a peptide, the fluorophore is presented at a slightly different orientation relative to the biomolecule.
While their fluorescence emission spectra are similar and both are effective for general labeling, research has demonstrated that 5-FAM and 6-FAM can display distinct trends in binding interactions and functional assays. For instance, studies on FAM-labeled DNA nanoantennas showed that despite similar fluorescence emission, the two isomers exhibited different quenching upon protein binding and different transient fluorescence spikes during enzymatic hydrolysis. Thus, for experiments where the fluorophore’s spatial orientation may influence target engagement, the choice of isomer should be carefully considered.
Some providers note that 5-FAM is a single isomer, whereas 6-FAM is often the default for custom oligonucleotide labeling due to its effective blockage of the 3′ terminus from polymerase extension. In peptide synthesis, both isomers are available, and many commercial facilities offer FAM in either form depending on the researcher’s preference.
FAM vs. FITC: What Is the Difference?
Another important distinction is between FAM (carboxyfluorescein) and FITC (fluorescein isothiocyanate). While both are green fluorophores with similar spectral properties, their conjugation chemistries differ. FAM typically contains a carboxylic acid group and is activated as an NHS ester for amine coupling. In contrast, FITC contains an isothiocyanate group that reacts directly with amines via a thiourea linkage. Observations suggest that FITC-labeled peptides tend to deteriorate more quickly than FAM conjugates, making FAM the preferred choice for long-term stability.
Find out more about peptide synthesis here.
Conjugation Chemistries and Protocols
NHS Ester Coupling to Primary Amines
The most common method for FAM conjugation is via NHS ester chemistry (5/6-carboxyfluorescein succinimidyl ester, FAM-NHS ester). In this approach, the NHS ester reacts efficiently with primary amines, such as the ε-amino group of lysine side chains or the N-terminal amine of the peptide, to form a chemically stable amide bond. Typical protocols involve adding the FAM-NHS ester to the peptide at a 4–20:1 molar ratio in a pH 8.0–8.5 buffer (e.g., phosphate buffer or sodium bicarbonate) and incubating at room temperature for 2–4 hours. The reaction is then quenched with Tris or glycine, and the labeled peptide is purified by HPLC.
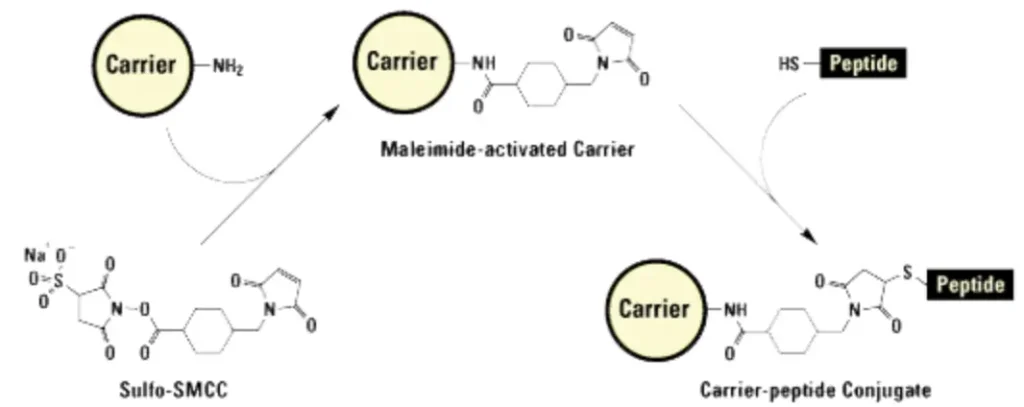
Maleimide Coupling to Thiols
For site-specific labeling of cysteine residues, FAM maleimide (6-isomer) is employed. The maleimide group reacts specifically with free sulfhydryl groups (-SH) at pH 6.5–7.5, forming a stable thioether bond. This method provides precise control over labeling position, as cysteines are less common in native sequences and can be strategically introduced during peptide design.
Spacers to Reduce Steric Hindrance
Given the bulky nature of the FAM fluorophore, incorporating a short spacer between the dye and the peptide core can mitigate steric interference and preserve biological activity. Common spacers include 6-aminohexanoic acid (Ahx) and β-alanine. LifeTein has described the use of Ahx spacers in N-terminal FITC-labeled peptides to achieve successful intracellular delivery for super-resolution imaging. Similarly, PEG-based spacers (e.g., PEG6) have been employed in FAM-labeled cell-penetrating peptides to maintain activity and enable cellular uptake.
Purification and Validation
Following conjugation, reverse-phase HPLC is used to separate the labeled peptide from unreacted dye and truncated products, achieving purities exceeding 95-98%. Analytical techniques such as mass spectrometry (MS) confirm the correct molecular weight of the conjugate, ensuring proper labeling.
Applications of FAM-Labeled Peptides
Live-Cell Imaging and Internalization Studies
FAM-labeled peptides are indispensable tools for tracking cellular uptake and intracellular trafficking. For example, FAM-conjugated ovalbumin peptide (Fam-ova, SIINFEKL) has been widely used to study MHC-I antigen presentation and visualize cytosolic antigen release via confocal microscopy. Similarly, FAM-labeled cell-penetrating peptides (e.g., TAT-derived conjugates) have enabled real-time monitoring of peptide internalization.
Flow Cytometry and Receptor Binding Assays
The green fluorescence of FAM is readily detected by standard flow cytometers, making it ideal for quantifying peptide-receptor interactions and competitive binding assays. FAM-labeled probes have been used to measure binding affinities in microscale thermophoresis (MST) and fluorescence polarization-based assays.
FRET-Based Assays and Enzyme Substrate Design
FAM also serves as an excellent donor fluorophore in FRET (Förster Resonance Energy Transfer) pairs. When paired with an appropriate acceptor dye, such as TAMRA or Dabcyl, FAM-labeled peptides enable real-time monitoring of protease activity, conformational changes, and molecular interactions within 1–10 nm distances. This approach is widely used in high-throughput screening and mechanistic enzymology.
Antibody and Immunoassay Development
Because FAM is a well-defined hapten, high-quality antibodies are commercially available for detecting FAM-labeled peptides in immunoassays. For instance, the FITC/5-FAM/6-FAM Rabbit mAb (A22444) recognizes both FAM isomers and offers high sensitivity for Western blotting, ELISA, and IHC applications.
Frequently Asked Questions (FAQ)
What is the difference between 5-FAM and 6-FAM?
The two are positional isomers of carboxyfluorescein, differing only in the location of the carboxylic acid group (C5 versus C6 position). While their fluorescence emission spectra are nearly identical, research has shown that they can exhibit different binding interactions in certain biological assays, such as protein binding and enzymatic hydrolysis. Therefore, the choice of isomer should be guided by the specifics of your experiment.
How do I choose between FAM and FITC for labeling?
FAM (carboxyfluorescein) and FITC (fluorescein isothiocyanate) share similar spectral properties, but their conjugation chemistries differ. FAM is typically activated as an NHS ester for amine coupling, whereas FITC reacts directly with amines via an isothiocyanate group. Notably, FAM-labeled peptides tend to be more stable than their FITC-conjugated counterparts. For long-term experiments requiring robust stability, FAM is therefore preferred.
Can I purchase custom FAM-labeled peptides for my research?
Yes. Specialized providers such as LifeTein offer custom synthesis of FAM-labeled peptides with high purity (>98%) and rigorous analytical validation. Their services include choices between 5-FAM and 6-FAM, optional spacers (Ahx, β-Ala), and a variety of conjugation positions (N-terminus, lysine side chain, cysteine-specific labeling).
What buffers should I use when working with FAM-labeled peptides?
FAM fluorescence is optimal between pH 7.5 and 8.5 and decreases sharply below pH 7. Therefore, neutral to slightly alkaline buffers (e.g., PBS at pH 7.4 or 8.0) are recommended for experiments involving FAM-labeled peptides. Stock solutions should be prepared in DMSO, as FAM is highly soluble in this organic solvent.
How should I store FAM-labeled peptides to maintain stability?
FAM-labeled peptides should be stored as lyophilized solids at -20°C, protected from light. Once reconstituted in aqueous buffer, they should be used promptly and kept on ice. Avoid repeated freeze-thaw cycles, as these may degrade the fluorescent signal.
References
Harroun, S. G., Lauzon, D., Ebert, M. C. C. J. C., Desrosiers, A., Wang, X., & Vallée-Bélisle, A. (2021). Monitoring protein conformational changes using fluorescent nanoantennas. Nature Methods, 19(1), 71–80. https://doi.org/10.1038/s41592-021-01355-5
Okuda-Shinagawa, N. M., Moskalenko, Y. E., Junqueira, H. C., Baptista, M. S., Marques, C. M., & Machini, M. T. (2017). Fluorescent and Photosensitizing Conjugates of Cell-Penetrating Peptide TAT(47-57): Design, Microwave-Assisted Synthesis at 60 °C, and Properties. ACS Omega, 2(11), 8156–8166. https://doi.org/10.1021/acsomega.7b01127