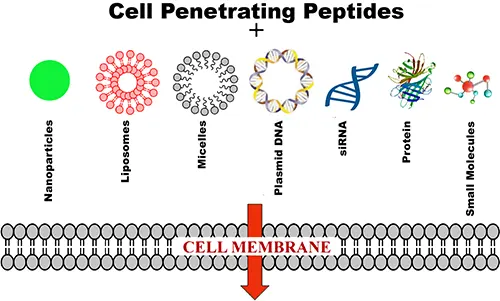
Cell-penetrating peptides (CPPs), also called cell-permeable peptides or protein transduction domains, are short peptide sequences used to improve intracellular delivery of otherwise membrane-limited cargo. Depending on the sequence and delivery format, CPPs can support transport of peptides, proteins, oligonucleotides, probes, nanoparticles, and related research cargos.
CPPs are not a single chemistry or a single mechanism. In practice, performance depends on peptide class, cargo type, linkage strategy, concentration, cell type, and the balance between endocytic and non-endocytic uptake. Arginine-rich and amphipathic CPPs are among the most widely studied formats in the literature, and they continue to be relevant in peptide, oligonucleotide, and nanoparticle delivery research.
| Main use | Improving intracellular delivery of peptides, proteins, oligonucleotides, and related cargos |
| Common CPP classes | Arginine-rich, lysine-rich, amphipathic, and sequence-derived transport peptides |
| Common formats | Standalone CPP synthesis, CPP–cargo conjugates, disulfide-linked delivery constructs, and non-covalent CPP–cargo complexes |
| Popular workflows | Cys(Npys)-R9 conjugation, Tat-derived peptides, penetratin-type peptides, stearylated polyarginine formats, and oligonucleotide delivery constructs |
| Related areas | Peptide–oligonucleotide conjugates, PNA conjugates, click chemistry conjugation, and LNP-facing peptide constructs |
1. Covalent CPP–cargo conjugates
In this format, the CPP is chemically attached to the cargo. This is often useful when a defined stoichiometry or a cleavable linkage is needed.
2. Disulfide-linked CPP constructs
A common practical strategy is to link the CPP to a cysteine-containing cargo through a disulfide bond, allowing intracellular release under reducing conditions.
3. Non-covalent CPP–cargo complexes
Some CPPs, especially arginine-rich formats, can form non-covalent complexes with nucleic acids or other negatively charged cargos and act as delivery carriers without permanent conjugation.
One of the most useful practical CPP formats is Cys(Npys)-(Arg)9: C(Npys)RRRRRRRRR-NH2. This peptide contains an activated cysteine residue with the Npys group, which can selectively react with a free thiol on a cysteine-containing peptide or cargo to form an unsymmetrical disulfide bond.
This type of construct can be useful when the delivery peptide should carry the cargo into the cell and then allow reductive release in the intracellular environment. This makes it relevant for peptide delivery and for certain nucleic acid or screening-oriented constructs.
We also provide related arginine-rich formats such as Stearyl-R8, FITC-Stearyl-R8, RRRRRRRRRC, and other CPP variants used in delivery studies.
Practical note
For many projects, the most useful service is not simply ordering a CPP sequence, but designing the correct CPP–cargo relationship: covalent vs non-covalent, cleavable vs non-cleavable, and peptide-only vs oligonucleotide or nanoparticle-facing format. For practical delivery examples, RNA mixing strategies, and product-linked CPP formats, see Cell-Penetrating Peptide Delivery Examples and Practical Formats.
| Name | Origin / Class | Representative Sequence |
| Tat (48–60) | HIV-1 derived | GRKKRRQRRRPPQ or related Tat-derived variants |
| Cys(Npys)-(Arg)9 | Arginine-rich delivery format | C(Npys)RRRRRRRRR-NH2 |
| Oligoarginine | Arginine-rich CPP | Rn |
| Transportan | Amphipathic chimeric CPP | GWTLNSAGYLLGKINLKALAALAKKIL |
| MPG-type peptides | gp41 / SV40-derived delivery peptides | GALFLGFLGAAGSTMGAWSQPKKKRKV |
| Pep-1 | Trp-rich amphipathic CPP | KETWWETWWTEWSQPKKKRRV |
| Penetratin | Antennapedia-derived | RQIKIWFQNRRMKWKK and related variants |
| Stearyl-R8 | Lipidated arginine-rich CPP | Stearyl-RRRRRRRR-amide |
CPP-mediated delivery has been especially explored for oligonucleotides, including siRNA and related cargos. In this area, both covalent conjugation and non-covalent complex formation have been studied. This is one reason CPPs are closely related to peptide–oligonucleotide conjugation and PNA delivery-related formats.
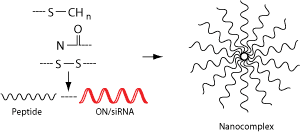
Stearylated polyarginine and related amphiphilic or arginine-rich CPPs are frequently used in nucleic-acid delivery research because the balance of cationic charge, hydrophobicity, and cargo association can be tuned according to the application.
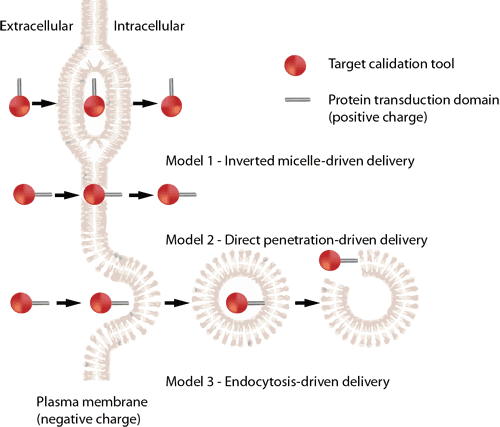
CPP uptake is not fully explained by one single mechanism. The older “direct translocation vs endocytosis” debate has evolved into a more nuanced view in which the same peptide may use different routes depending on conditions such as concentration, membrane composition, cargo type, and cell context. Reviews continue to describe both endocytic uptake and non-endocytic translocation-related behavior, especially for arginine-rich CPPs.
| CPP | Why it matters for service work |
| HIV Tat-derived peptides | Widely recognized CPP class; often used as a reference format for intracellular delivery studies |
| Polyarginine formats such as R8 or R9 | Practical for peptide or oligonucleotide delivery designs; often useful in conjugation or complex-formation studies |
| Cys(Npys)-R9 | Especially useful when a cysteine-containing cargo needs a selective disulfide-linked delivery format |
| Penetratin / Antennapedia-derived peptides | Common amphipathic / sequence-derived CPP reference class |
| Stearylated arginine-rich peptides | Useful when additional hydrophobicity helps tune delivery behavior |
Download the full list: Cell-penetration peptide list