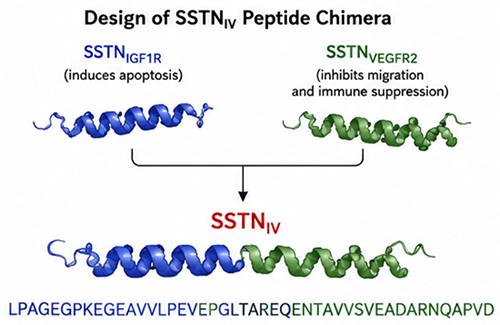
The peptide sequence LPAGEGPKEGEAVVLPEVEPGLTAREQENTAVVSVEADARNQAPVD corresponds to the engineered SSTNIV peptide chimera reported in advanced multiple myeloma research as a multifunctional inhibitor targeting syndecan-1 (Sdc1/CD138)-dependent signaling pathways. The peptide was synthesized by LifeTein and evaluated in mechanistic and therapeutic studies investigating tumor progression, immune suppression, and bone marrow microenvironment remodeling in aggressive myeloma models.
Unlike conventional peptide inhibitors that target a single pathway, SSTNIV was rationally designed as a dual-function peptide capable of simultaneously disrupting:
- Sdc1-mediated survival signaling that protects myeloma cells from apoptosis
- Sdc1-mediated migratory and immunosuppressive signaling that promotes tumor progression within the bone marrow microenvironment
This dual-targeting mechanism makes SSTNIV particularly important for studies examining the complex interactions between tumor cells, stromal cells, macrophages, T cells, and extracellular matrix components in relapsed and refractory multiple myeloma.
Scientific Background: Syndecan-1 in Multiple Myeloma
Syndecan-1 (CD138) is a transmembrane heparan sulfate proteoglycan highly expressed on plasma cells and multiple myeloma cells. Sdc1 functions as a signaling organizer that coordinates receptor tyrosine kinases, integrins, extracellular matrix interactions, and growth factor signaling.
In multiple myeloma, Sdc1 contributes to:
- Myeloma cell survival and resistance to apoptosis
- Cell migration and invasion
- Tumor-associated macrophage recruitment
- Immune suppression within the bone marrow microenvironment
- Angiogenesis and metastatic dissemination
- Resistance to chemotherapy and proteasome inhibitors
Because of its central role in organizing pro-tumor signaling complexes, Sdc1 has emerged as an attractive therapeutic target in hematologic malignancies.
Engineering Strategy Behind SSTNIV
SSTNIV was engineered as a chimeric peptide by combining active domains from two previously characterized syndecan-targeting peptides:
- SSTNIGF1R — disrupts Sdc1-IGF1R-integrin signaling and induces apoptosis
- SSTNVEGFR2 — disrupts Sdc1/VEGFR2/VLA-4 signaling involved in migration and immune suppression
The resulting SSTNIV chimera was designed to simultaneously inhibit tumor survival pathways and remodel the immunosuppressive tumor microenvironment.
Major Findings Reported in the Publication
1. Direct Anti-Myeloma Activity
The publication demonstrated that SSTNIV directly induced apoptosis in multiple myeloma cells by disrupting syndecan-1-associated receptor signaling complexes required for tumor survival.
The peptide inhibited:
- IGF1R signaling
- Integrin-associated survival pathways
- Migration-associated VEGFR2 signaling
- Bone marrow niche interactions supporting tumor persistence
2. Suppression of Tumor Cell Migration
SSTNIV significantly reduced migration and invasion of myeloma cells. The study demonstrated that the peptide interfered with signaling pathways associated with VLA-4 and VEGFR2, both of which are important for trafficking of myeloma cells within the bone marrow microenvironment.
Because metastatic dissemination and bone marrow colonization contribute to disease progression, inhibition of tumor cell motility is considered an important therapeutic feature.
3. Reversal of Immune Suppression
One of the most important findings of the study was the ability of SSTNIV to reverse multiple forms of immune suppression associated with advanced myeloma.
The peptide:
- Reduced tumor-associated macrophage (TAM) accumulation
- Reduced regulatory T-cell (Treg) infiltration
- Enhanced cytotoxic CD8+ T-cell migration
- Improved anti-tumor immune activity within the marrow
This is particularly significant because the myeloma microenvironment is highly immunosuppressive and frequently limits the effectiveness of anti-cancer immune responses.
4. In Vivo Anti-Tumor Activity
In aggressive VQ murine multiple myeloma models, SSTNIV treatment:
- Reduced tumor burden
- Reduced bone marrow disease progression
- Decreased extramedullary tumor dissemination
- Improved bone marrow cellularity
- Significantly prolonged survival
Combination therapy studies further demonstrated enhanced efficacy when SSTNIV was combined with the proteasome inhibitor bortezomib.
5. Improved Stability Compared with Parent Peptides
The study also reported that SSTNIV exhibited significantly improved in vivo stability compared with one of the parent peptides. The peptide demonstrated a reported circulation half-life of approximately 46.5 hours in mice, supporting prolonged biological activity and sustained therapeutic effects.
Applications in Biomedical Research
- Multiple myeloma biology research
- Syndecan-1 (CD138) signaling studies
- Tumor microenvironment remodeling research
- Immune suppression and immunotherapy studies
- Tumor-associated macrophage (TAM) biology
- T-cell trafficking and activation studies
- Peptide-based anti-cancer therapeutic development
- Combination therapy studies with proteasome inhibitors
Why SSTNIV Is Important
Most anti-cancer peptides target only a single tumor-associated pathway. SSTNIV is particularly important because it simultaneously targets:
- Tumor survival signaling
- Tumor migration and invasion
- Immune suppression within the tumor microenvironment
- Bone marrow niche interactions
This multifunctional mechanism makes SSTNIV a valuable research tool for studying how syndecan-organized receptor complexes regulate both cancer progression and immune evasion.
The peptide also highlights the growing role of engineered multifunctional peptides in oncology research, where synthetic peptide chimeras are increasingly being developed to target complex signaling networks that cannot be effectively blocked by single-pathway inhibitors.
Publication
Jung O, Beauvais DLM, Ibaan GL, et al.
SSTNIV, a syndecan-1-targeting peptide chimera, reverses immune suppression and inhibits myeloma progression.
Signal Transduction and Targeted Therapy. 2026;11:175.
DOI: 10.1038/s41392-026-02709-1
The publication reported that the SSTNIV peptide used in the study was synthesized by LifeTein.
Read the publication here:
Nature Publication
Related Services
Custom peptide synthesis
Long peptide synthesis
Peptide conjugation and click chemistry
Fluorescent peptide labeling
Research Use Only. Not for human or therapeutic use.
|