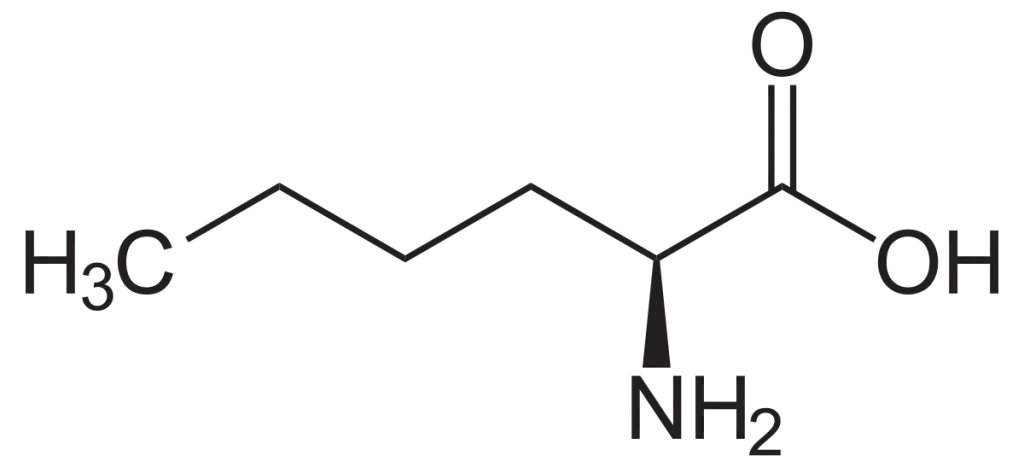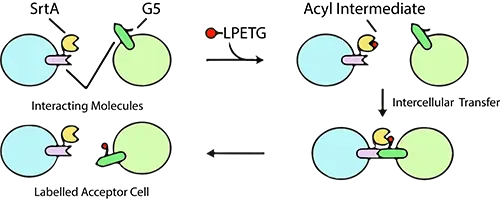
The study of protease activity is critical for understanding cellular processes, disease mechanisms, and drug development. Among the various tools available for protease research, the Biotin-Ahx-LPETGS-NH2 substrate has emerged as a highly specific and versatile reagent. This peptide substrate is designed to detect and quantify the activity of sortase A, an enzyme widely used in protein engineering and bioconjugation. In this article, we explore the structure, applications, and significance of the Biotin-Ahx-LPETGS-NH2 substrate.
Key Takeaways
- Biotin-Ahx-LPETGS-NH2 is a peptide substrate specifically designed for sortase A activity assays.
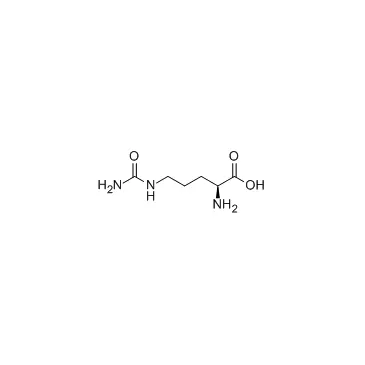
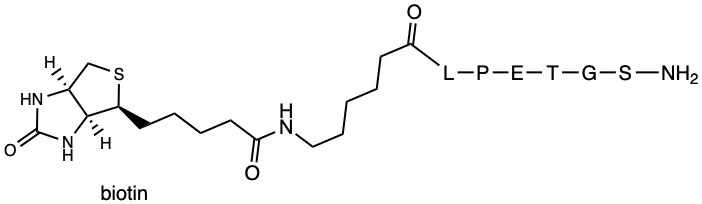
- The substrate features a biotin tag for easy detection and purification, an Ahx (6-aminohexanoic acid) spacer for flexibility, and the LPETGS recognition sequence for sortase A.
- It is widely used in protein labeling, site-specific protein modification, and enzyme kinetics studies.
- The substrate’s design enables high sensitivity and specificity in detecting sortase A activity.
- Applications include bioconjugation, live-cell imaging, and drug discovery.
Structure and Design of Biotin-Ahx-LPETGS-NH2
Biotin Tag for Detection and Purification
The biotin tag in the Biotin-Ahx-LPETGS-NH2 substrate serves as a universal handle for detection and purification. Biotin’s strong affinity for streptavidin allows for efficient immobilization on solid supports or visualization using streptavidin-conjugated fluorophores. This feature is particularly useful in ELISA, Western blotting, and pull-down assays, where the substrate’s interaction with sortase A can be easily monitored.
Ahx Spacer for Enhanced Flexibility
The inclusion of an Ahx (6-aminohexanoic acid) spacer between the biotin tag and the LPETGS sequence provides structural flexibility. This spacer ensures that the biotin tag does not sterically hinder the interaction between the substrate and sortase A, thereby maintaining high enzymatic efficiency. Additionally, the Ahx spacer improves the solubility of the peptide, making it suitable for a wide range of experimental conditions.
LPETGS Recognition Sequence
The LPETGS sequence is the core recognition motif for sortase A, a transpeptidase enzyme derived from Staphylococcus aureus. Sortase A cleaves the peptide bond between the threonine (T) and glycine (G) residues, enabling the attachment of functional groups or proteins to the C-terminus of the substrate. This sequence-specific cleavage is the basis for the substrate’s high specificity in sortase A activity assays.
Find our flagship LPETGS product here.
Applications of Biotin-Ahx-LPETGS-NH2
Protein Labeling and Bioconjugation
One of the primary applications of the Biotin-Ahx-LPETGS-NH2 substrate is in protein labeling and bioconjugation. Sortase A-mediated reactions allow for the site-specific attachment of labels, such as fluorophores or affinity tags, to proteins of interest. This approach is widely used in antibody-drug conjugates (ADCs), fluorescent protein tagging, and surface immobilization for biosensors.
Enzyme Kinetics Studies
The substrate is also employed in enzyme kinetics studies to characterize the activity and specificity of sortase A. By monitoring the cleavage of the LPETGS sequence, researchers can determine kinetic parameters such as Km and kcat. These studies provide valuable insights into the catalytic mechanism of sortase A and its potential applications in protein engineering.
Live-Cell Imaging
In live-cell imaging, the Biotin-Ahx-LPETGS-NH2 substrate can be used to visualize protease activity in real-time. The biotin tag allows for the incorporation of fluorescent probes, enabling the detection of sortase A activity in living cells. This application is particularly useful for studying cell surface dynamics and protein-protein interactions in their native environment.
Advantages of Biotin-Ahx-LPETGS-NH2
High Sensitivity and Specificity
The Biotin-Ahx-LPETGS-NH2 substrate offers high sensitivity and specificity for sortase A, making it an ideal tool for detecting low levels of enzyme activity. The LPETGS sequence ensures that the substrate is exclusively cleaved by sortase A, minimizing off-target effects.
Versatility in Experimental Design
The substrate’s modular design allows for customization to suit specific experimental needs. For example, the biotin tag can be replaced with other affinity tags or fluorophores, depending on the application. This versatility makes the substrate a valuable reagent in both basic research and industrial applications.
Find our other LPETG products here.
Compatibility with High-Throughput Assays
The Biotin-Ahx-LPETGS-NH2 substrate is compatible with high-throughput screening (HTS) platforms, enabling the rapid identification of sortase A inhibitors or activators. This capability is particularly relevant in drug discovery, where the substrate can be used to screen large compound libraries for potential therapeutic agents.
Pasqual, G., Chudnovskiy, A., Tas, J. et al. Monitoring T cell–dendritic cell interactions in vivo by intercellular enzymatic labelling. Nature 553, 496–500 (2018). https://doi.org/10.1038/nature25442