LifeTein provides fluorescent peptide modification services for a wide range of research applications, including fluorescence microscopy, flow cytometry, intracellular tracking, live-cell imaging, FRET assays, and enzyme studies. Available labels include FITC, FAM, TAMRA, cyanine dyes, Alexa Fluor dyes, ATTO dyes, EDANS/Dabcyl, and other fluorescent or quencher formats.
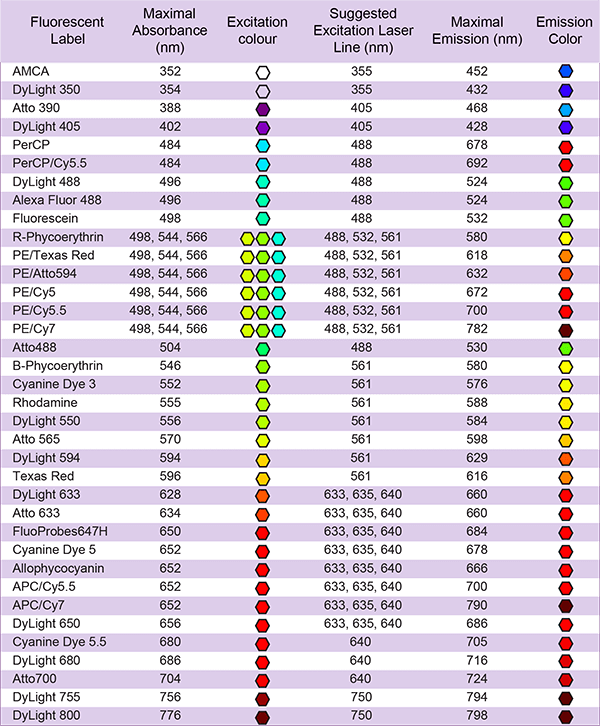
| Common dyes | FITC, FAM, AMCA, TAMRA, Cy3, Cy3.5, Cy5, Cy5.5, Cy7, Cy7.5, MCA, EDANS/Dabcyl, AZDye, BODIPY FL, Alexa Fluor, ATTO dyes, and more |
| Applications | FRET assays, fluorescence microscopy, flow cytometry, intracellular delivery studies, localization studies, and enzyme substrate design |
| Labeling options | N-terminus, selected side-chain positions such as Lys or Cys, and peptide designs with optional spacers such as Ahx or β-Ala |
| Peptide formats | Fluorescent peptides, quenched substrates, donor-acceptor FRET peptides, and selected near-infrared peptide labels |
| Support | Technical review for dye selection, spacer recommendations, and sequence-specific feasibility |
LifeTein provides these fluorescent labeling options: FITC, FAM, AMCA, TAMRA, Cyanine Dye Cy3, Cy3.5, Cy5, Cy5.5, Cy7, Cy7.5, EDANS/Dabcyl, MCA, AZDye, BODIPY FL, Alexa Fluor dyes (Alexa488, Alexa532, Alexa546, Alexa594, Alexa633, Alexa647), ATTO dyes (Atto465, Atto488, Atto495, Atto532, Atto550, Atto647), and DyLight dyes (DyLight 488, DyLight 550). Contact us if your desired dye is not listed.
Reference:
...FITC-Tet1, Alexa 594-N1, and Atto 643-N1 (95% purity) were acquired from LifeTein (NJ)...
AZDye or Alexa Fluor 647 labeling, Cy3, Cy5, and Cy7 labeling (Structurally, AZDye 647 and Alexa Fluor 647 have similar fluorophores. Spectrally, Alexa Fluor 647, AZDye 647, CF 647 dye, Cy5 dye, and other Cyanine5-based dyes are closely related.)
QSY 9 succinimidyl ester labeling
QSY 9 succinimidyl ester has strong visible absorption around 560 nm but is essentially nonfluorescent. It is commonly used as an acceptor in fluorescence resonance energy transfer (FRET) applications.
Fluorescent dyes are among the most widely used labels for biomolecules. FITC derivatives remain common and versatile tools for fluorescence microscopy, flow cytometry, and immunofluorescence-based assays.

Need help selecting a fluorescent dye or spacer?
Send us your peptide sequence, intended assay, target wavelength, and preferred dye. We can help review practical labeling options for your project.
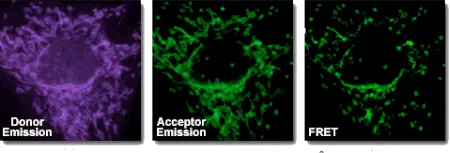
Fluorescence resonance energy transfer (FRET) is a method that detects distance-dependent interactions between two dye-linked molecules. Energy is transferred from a donor to an acceptor without emission of a photon. FRET assays are rapid, sensitive, and practical for many biomolecular studies. Although a single dye can sometimes be used, most applications require a donor-acceptor pair.
A practical measure of FRET efficiency is the Förster radius, the distance at which the rate of energy transfer equals the rate of donor fluorescence. FRET is particularly useful for studying co-localization events in biological samples, especially in live-cell imaging.
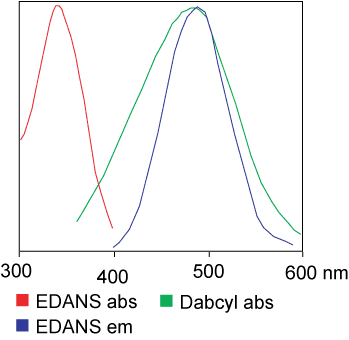
Energy transfer depends on spectral overlap between donor emission and acceptor absorption and usually occurs when donor and acceptor dyes are within about 10–100 Å.
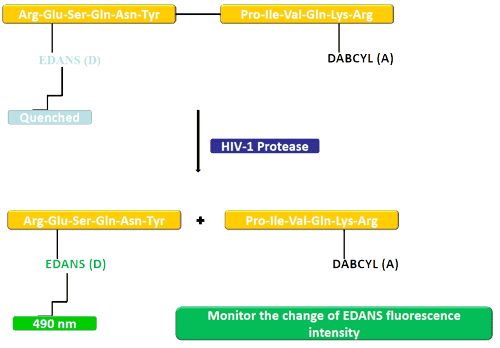
In this example, the donor (EDANS) and quencher (DABCYL) are attached to a substrate of HIV protease. In the uncleaved substrate, DABCYL quenches EDANS; after cleavage, EDANS fluorescence can be detected.
FRET peptides are useful for studying peptidase specificity because they allow reactions to be monitored continuously and enzyme activity to be measured rapidly. Cleavage between donor and acceptor can generate a measurable fluorescent signal that permits nanomolar levels of enzymatic activity to be detected.
FRET peptides are used in many enzyme-related studies, including:
Standard dye combinations used for FRET:
Useful FRET calculator: www.fpbase.org/fret/
Donor-acceptor pairs capable of quenching by resonance energy transfer in peptide substrates of proteolytic enzymes
|
Wavelengths (nm) |
||
Quencher |
Fluorophore |
Excitation |
Emission |
Dabcyl |
Edans |
336 |
490 |
The Förster Critical Distance for Common RET Donor-Acceptor Pairs
Donor |
Acceptor |
Förster Distance |
Trp |
Dansyl |
2.1 |
Name |
Ex/Em (nm) |
Emission color |
|---|---|---|
Abz, Anthranilyl, 2-Aminobenzoyl |
320/420 |
Blue |
MCA, 7-Methoxycoumarinyl-4-acetyl |
328/393 |
Blue |
AMCA, Fmoc-Lys(AMCA)-OH |
345/450 |
Blue |
Alexa Fluor dyes: Alexa488, Alexa532, Alexa546, Alexa594, Alexa633, Alexa647 |
650/668 |
Blue / Red (depending on dye) |
FITC or BODIPY FL |
494/521 |
Green |
FAM |
494/520 |
Green |
Cy3 |
555/570 |
Yellow |
TAMRA |
556/563 |
Yellow |
ATTO dyes: Atto465, Atto488, Atto495, Atto532, Atto550, Atto647 |
453/508 |
Varies by dye |
Cy3.5 |
591/604 |
Orange |
Texas Red |
589/615 |
Red |
Cy5, Alexa Fluor 647, AZDye 647, CF 647 |
646/662 |
Red |
Cy5.5 |
673/707 |
Near-infrared |
DyLight dyes: DyLight 488, DyLight 550 |
493/518 |
Varies by dye |
Cy7 |
750/773 |
Near-infrared |
Cy7.5 |
788/808 |
Near-infrared |
Reference:
Fluorescent peptide Angiotensin 1-7 (Ang-(1-7)) with the FRET pair Abz/[Tyr7(NO2)] was synthesized by LifeTein. This peptide was used to study the Ang-(1-7) endopeptidase in the renal renin angiotensin system.
...100 µM Abz-Ang-(1-7)-[Tyr7(NO2)], an internally quenched fluorescent peptide (synthesized by LifeTein, South Plainfield, NJ, USA)...
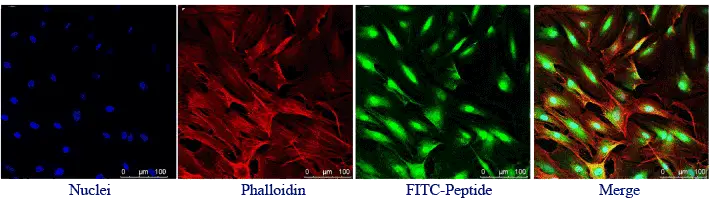
FITC can react with sulfhydryl groups, such as reduced cysteine side chains, and can also be introduced through primary amino groups depending on peptide design.
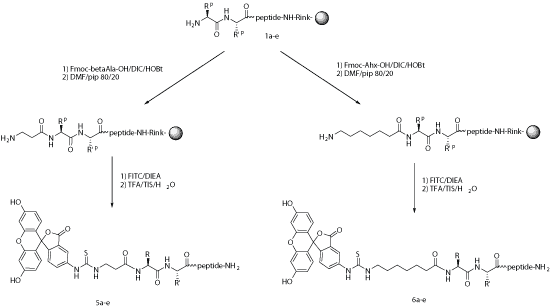
For many applications, fluorescent labels can be introduced during chemical synthesis. After selective deprotection, FITC may react with lysine, ornithine, or a primary amino group at the N-terminus of the growing peptide. In the N-terminal format, a spacer such as aminohexanoic acid (Ahx) is often introduced between the peptide and the dye.
Under acidic cleavage conditions, N-terminal FITC-labeled peptides can undergo cyclization that may lead to loss of the terminal amino acid. This can often be reduced or avoided by using a spacer such as Ahx or by using non-acidic cleavage strategies where appropriate.
Ahx or β-Ala can be used successfully as spacers to reduce steric hindrance and improve fluorescent labeling performance in FITC-labeled peptides.
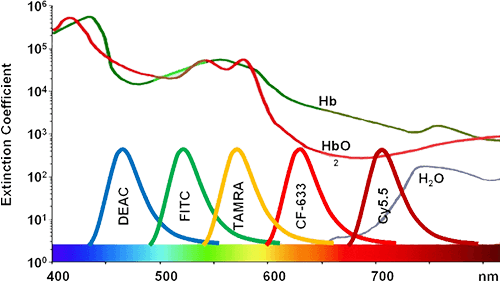
Tetramethylrhodamine (TAMRA) is another useful fluorophore and offers several advantages over FITC in selected intracellular applications. TAMRA is more resistant to photobleaching, has different excitation and emission maxima, and may provide more uniform intracellular distribution. Similar to Cy3, TAMRA is stable and bright. FITC and TAMRA can also be used together because their excitation and emission maxima are sufficiently separated and they do not strongly quench one another.
BODIPY FL is a bright, photostable green fluorescent dye with excitation and emission similar to FITC, FAM, or Alexa Fluor 488. It is a useful choice for microscopy and fluorescence polarization assays.
Fluorescent dyes such as biotin and FITC can generally be introduced at either the N- or C-terminus of a peptide. In many peptide projects, N-terminal modification is preferred because it typically offers shorter turnaround time, higher success probability, and fewer extra coupling steps.
Because many dyes are large aromatic molecules, adding a flexible spacer such as Ahx can help reduce steric interference between the fluorophore and the peptide, helping preserve peptide conformation and biological activity. FITC can also be linked to cysteine thiols or lysine amino groups.
| Probe | Ex (nm) | Em (nm) | MW | Notes |
|---|---|---|---|---|
| Methoxycoumarin (MCA) | 360 | 410 | 317 | Succinimidyl ester |
| FITC | 495 | 519 | 389 | pH sensitive |
| X-Rhodamine | 570 | 576 | 548 | |
| Rhodamine B | 570 | 590 | ||
| Cy2 | 492 | 510 | ||
| Cy3 | 550 | 570 | ||
| Cy5 | 650 | 670 |
ATTO Dyes
ATTO dyes can be used as alternatives to more traditional fluorescent labels. They offer enhanced photostability, improved ozone resistance, long signal lifetimes, and lower background for higher sensitivity. They are particularly useful in multiplex applications using visible and near-IR wavelengths.
For example, Atto 488 is often considered an alternative to FITC or Alexa Fluor 488, while Atto 550 may serve as an alternative to rhodamine dyes, Cy3, and Alexa Fluor 550.
| Fluorophores | Recommended ATTO Dye Alternative |
|---|---|
| Alexa Fluor 488 | Atto 488 |
| FITC | Atto 488 |
| FAM | Atto 488 |
| JOE | Atto 520 |
| TET | Atto 520 |
| Alexa Fluor 532 | Atto 532 |
| HEX™ | Atto 532, Atto Rho6G |
| TAMRA | Atto 550 |
| Cy3 | Atto 550 |
| Cy3.5 | Atto 565 |
| ROX | Atto 565, Atto Rho11 |
| Alexa Fluor 594 | Atto 590, Atto 594 |
| Texas Red | Atto 590 |
| Alexa Fluor 633 | Atto 633, Atto Rho14 |
| Cy5 | Atto 647, Atto 647N, Atto 655 |
| Alexa Fluor 647 | Atto 647, Atto 647N, Atto 655 |
| Cy5.5 | Atto 680, Atto 700 |
| Light source | Main lines (nm) | Recommended ATTO dyes |
|---|---|---|
| Mercury arc lamp | 365, 405, 436, 546 | Atto 390, Atto 425, Atto 465, Atto 550, Atto 565 |
| Mercury arc lamp | 577 | Atto 590, Atto Rho101, Atto 594, Atto Rho13, Atto 610, Atto 611x |
| Xenon arc lamp | Continuum and peaks >800 nm | Atto 610, Atto 620, Atto 647, Atto 647N, Atto 655, Atto 680 |
| Halogen lamp | Low UV/violet emission; higher intensity at longer wavelengths | Atto 610, Atto 620, Atto 647, Atto 647N, Atto 655, Atto 680 |
| Argon ion laser | 488, 514 | Atto 488, Atto 520, Atto 532, Atto 550 |
| Argon-krypton laser | 488, 514, 647, 676 | Atto 520, Atto 647, Atto 647N, Atto 655, Atto 680 |
| Krypton laser | 647, 676 | Atto 647, Atto 647N, Atto 655, Atto Oxa12, Atto 665, Atto 680, Atto 700, Atto 725, Atto 740 |
| He-Ne laser | 633 | Atto Rho14, Atto 633, Atto 647, Atto 647N |
| Nd-YAG laser | 532 | Atto 532, Atto Rho6G, Atto 550, Atto 565, Atto Rho11, Atto Rho12 |
| Common diode laser | 635, 650, 670 | Atto 633, Atto 647, Atto 647N, Atto 655, Atto 680 |
Förster radius information for selected ATTO dye pairs is shown below.

Please click here to see more FAQs
Is a spacer required for fluorescent modification?
How should I dissolve peptides?
How do I choose the best level of peptide purity for my research?
This case study shows a commonly used FRET design. The fluorescent dye MCA (methoxycoumarin acetic acid) was incorporated at the N-terminus of a peptide substrate for stromelysin, a matrix metalloprotease. The quencher N-3-(2,4-dinitrophenyl)-L-2,3-diamino propionyl (DPA) was incorporated so that cleavage of the Gly-Leu bond separated donor and quencher.
The sequence used was:
MCA-Pro-Leu-Gly-Leu-DPA-Ala-Arg-NH2
MCA has peak excitation and emission at 328 nm and 393 nm, respectively. DPA has strong absorption at 363 nm with a shoulder at 410 nm, overlapping sufficiently with MCA fluorescence to allow quenching. Cleavage separates the two groups and leads to a marked increase in fluorescence.
This fluorescence increase can be used to monitor reaction rate and determine kinetic parameters for matrix metalloproteases, as well as to evaluate inhibitor potency.
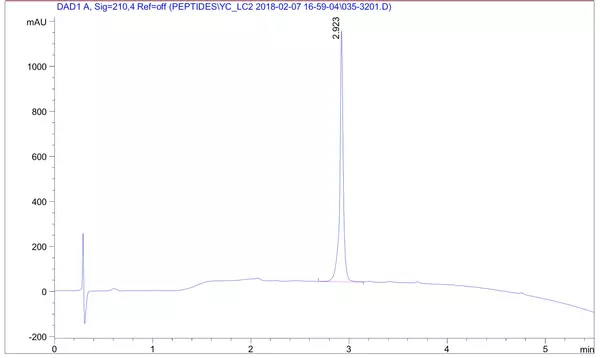
A client requested a very hydrophobic peptide 68 amino acids in length at 85% purity with FITC modification at the N-terminus. The peptide was synthesized successfully in 4 weeks.
HPLC Results:
MS Results: