LifeTein provides fluorescent peptide modification services for fluorescence microscopy, flow cytometry, intracellular tracking, live-cell imaging, FRET assays, enzyme substrate studies, peptide localization, and binding assays. Available labels include FITC, FAM, TAMRA, Cy3, Cy5, Cy7, Alexa Fluor dyes, ATTO dyes, EDANS/Dabcyl, MCA/DNP, and other fluorescent or quencher formats.
| Common dyes | FITC, FAM, AMCA, TAMRA, Cy3, Cy3.5, Cy5, Cy5.5, Cy7, Cy7.5, MCA, EDANS/Dabcyl, AZDye, BODIPY FL, Alexa Fluor, ATTO dyes, DyLight dyes, and more |
| Applications | FRET assays, fluorescence microscopy, flow cytometry, intracellular delivery studies, localization studies, enzyme substrate design, and peptide-protein interaction studies |
| Labeling positions | N-terminus, C-terminus, Lys side chain, Cys side chain, and other sequence-dependent positions |
| Spacer options | Ahx, β-Ala, PEG spacers, and other linkers to improve dye accessibility and reduce steric interference |
| Peptide formats | Fluorescent peptides, quenched substrates, donor-acceptor FRET peptides, near-infrared labeled peptides, cyclic fluorescent peptides, and dye-labeled cell-penetrating peptides |
| Technical support | Dye selection, spacer recommendation, labeling-position review, and sequence-specific feasibility evaluation |
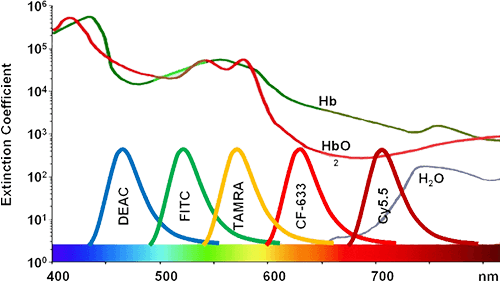
Select a dye to view excitation/emission spectra and recommended laser compatibility.
Dashed line = excitation, solid line = emission. Vertical markers indicate common laser compatibility for the selected dye.
Ex/Em: 495/519 nm
Molecular Weight: 389
Laser: 488 nm
Filters: 530/30
Description:
Classic green fluorophore widely used for peptide labeling, microscopy, flow cytometry, and intracellular tracking.
Typical Applications:
Need help selecting a fluorescent dye or spacer?
Send us your peptide sequence, intended assay, target wavelength, and preferred dye. We can help review practical labeling options for your project.
Use the sections below to compare dye options, review FRET peptide designs, understand labeling chemistry, and view representative LifeTein case studies.
Dye selection depends on the assay format, excitation source, emission channel, desired brightness, photostability, peptide solubility, and whether the peptide will be used in live cells, fixed cells, purified biochemical assays, or in vivo imaging models.
| Dye / Label | Typical Ex/Em | Best Use | Notes |
|---|---|---|---|
| FITC | 495/519 nm | Green fluorescence, microscopy, flow cytometry | Classic, economical, pH-sensitive |
| FAM | 494/520 nm | Green fluorescent peptides, FRET substrates | Common alternative to FITC |
| TAMRA | 556/563 nm | Orange-red fluorescence, imaging, FRET | More photostable than FITC in many applications |
| Cy3 | 555/570 nm | Orange fluorescence, microscopy, labeling | Bright cyanine dye |
| Cy5 / Alexa Fluor 647 / ATTO 647N | ~646/662 nm | Far-red peptide labeling | Useful for lower autofluorescence and multiplexing |
| Cy7 / Cy7.5 | ~750–788 / 773–808 nm | Near-infrared labeling | Useful when longer wavelength detection is required |
| MCA / AMC / Abz | UV-blue range | Enzyme substrates and FRET peptides | Often paired with DNP, Dabcyl, or Tyr(NO2) |
| EDANS / Dabcyl | Donor / quencher pair | Internally quenched FRET substrates | Common for protease substrate design |
ATTO dyes are useful alternatives to traditional fluorophores because they often provide strong brightness, good photostability, low background, and compatibility with common laser lines.
| Fluorophore | Recommended ATTO Alternative |
|---|---|
| Alexa Fluor 488 | ATTO 488 |
| FITC | ATTO 488 |
| FAM | ATTO 488 |
| JOE / TET | ATTO 520 |
| Alexa Fluor 532 | ATTO 532 |
| HEX | ATTO 532, ATTO Rho6G |
| TAMRA | ATTO 550 |
| Cy3 | ATTO 550 |
| Cy3.5 | ATTO 565 |
| ROX | ATTO 565, ATTO Rho11 |
| Alexa Fluor 594 | ATTO 590, ATTO 594 |
| Texas Red | ATTO 590 |
| Alexa Fluor 633 | ATTO 633, ATTO Rho14 |
| Cy5 | ATTO 647, ATTO 647N, ATTO 655 |
| Alexa Fluor 647 | ATTO 647, ATTO 647N, ATTO 655 |
| Cy5.5 | ATTO 680, ATTO 700 |
| Light Source | Main Lines | Recommended ATTO Dyes |
|---|---|---|
| Mercury arc lamp | 365, 405, 436, 546 nm | ATTO 390, ATTO 425, ATTO 465, ATTO 550, ATTO 565 |
| Mercury arc lamp | 577 nm | ATTO 590, ATTO Rho101, ATTO 594, ATTO Rho13, ATTO 610, ATTO 611x |
| Xenon arc lamp | Continuum and peaks >800 nm | ATTO 610, ATTO 620, ATTO 647, ATTO 647N, ATTO 655, ATTO 680 |
| Argon ion laser | 488, 514 nm | ATTO 488, ATTO 520, ATTO 532, ATTO 550 |
| Argon-krypton laser | 488, 514, 647, 676 nm | ATTO 520, ATTO 647, ATTO 647N, ATTO 655, ATTO 680 |
| He-Ne laser | 633 nm | ATTO Rho14, ATTO 633, ATTO 647, ATTO 647N |
| Nd-YAG laser | 532 nm | ATTO 532, ATTO Rho6G, ATTO 550, ATTO 565, ATTO Rho11, ATTO Rho12 |
| Common diode laser | 635, 650, 670 nm | ATTO 633, ATTO 647, ATTO 647N, ATTO 655, ATTO 680 |
Förster radius information for selected ATTO dye pairs is shown below.

Fluorescence resonance energy transfer, or FRET, is a distance-dependent fluorescence method used to study molecular proximity, peptide cleavage, enzyme activity, conformational change, and protein-protein association. In a typical FRET peptide substrate, a donor fluorophore and acceptor dye or quencher are placed on the same peptide. When the peptide is intact, fluorescence is quenched or transferred. After enzymatic cleavage or conformational change, the fluorescence signal changes.
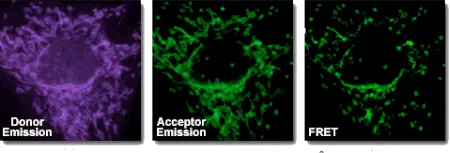
Energy transfer depends on spectral overlap between donor emission and acceptor absorption, donor-acceptor orientation, and distance. FRET is most useful when the donor and acceptor are within approximately 10–100 Å.
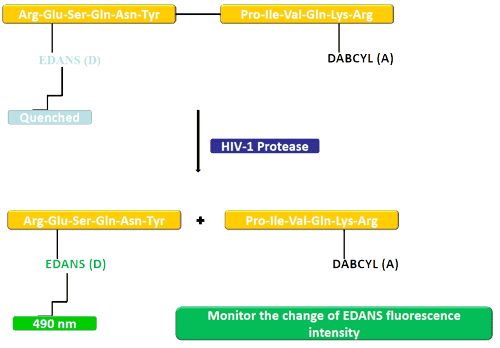
Example: donor EDANS and quencher DABCYL are attached to a protease substrate. In the uncleaved peptide, DABCYL quenches EDANS. After cleavage, EDANS fluorescence can be detected.
Useful FRET calculator: FPbase FRET Calculator
| Quencher | Fluorophore | Excitation | Emission |
|---|---|---|---|
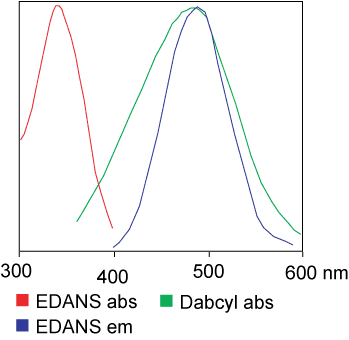
| Dabcyl | EDANS | 336 nm | 490 nm |
| Dansyl | Trp | 336 nm | 350 nm |
| DNP | MCA | 328 nm | 393 nm |
| Tyr(NO2) | Abz | 320 nm | 420 nm |
| Donor | Acceptor | Förster Distance |
|---|---|---|
| Trp | Dansyl | 2.1 nm |
| Dansyl | FITC | 3.3–4.1 nm |
| Dansyl | Rhodamine | 4.3 nm |
| FITC | Tetramethylrhodamine | 4.9–5.5 nm |
| Cy3 | Cy5 | >5.0 nm |
Fluorescent peptide Angiotensin 1-7 with the FRET pair Abz/[Tyr7(NO2)] was synthesized by LifeTein. This peptide was used to study Ang-(1-7) endopeptidase activity in the renal renin angiotensin system.
“100 µM Abz-Ang-(1-7)-[Tyr7(NO2)], an internally quenched fluorescent peptide (synthesized by LifeTein, South Plainfield, NJ, USA)…”
Fluorescent dyes can be introduced at the N-terminus, C-terminus, Lys side chain, Cys side chain, or other engineered positions depending on the peptide sequence and application. The best labeling strategy depends on whether the dye must avoid the active binding motif, whether the peptide requires a free terminus, and whether a spacer is needed.
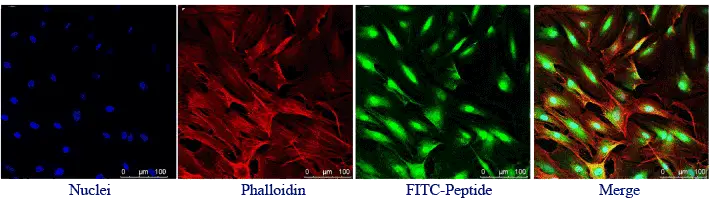
FITC is one of the most widely used green fluorescent labels for peptide synthesis. It can be introduced through primary amino groups such as the N-terminus or selected Lys side chains, and it may also be incorporated through cysteine-based strategies depending on the dye format.
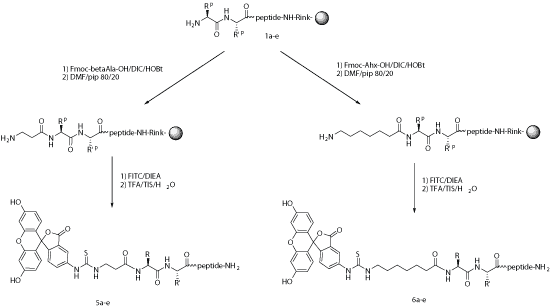
In many N-terminal FITC peptide designs, a spacer such as aminohexanoic acid (Ahx) is recommended between the dye and the peptide sequence. This helps reduce steric hindrance and may preserve peptide binding, receptor interaction, or cellular uptake.
Under strongly acidic cleavage conditions, N-terminal FITC-labeled peptides can sometimes undergo side reactions or cyclization-related loss of the terminal amino acid. This risk can often be reduced by using an Ahx or β-Ala spacer and by reviewing the sequence before synthesis.
Because fluorescent dyes are often bulky aromatic molecules, a spacer can help separate the dye from the peptide’s active region.
TAMRA is often used when a red-shifted label is desired. Compared with FITC, TAMRA may provide better photostability in selected applications and can be useful for intracellular imaging, microscopy, and FRET designs.
BODIPY FL is a bright, photostable green fluorescent dye with excitation and emission similar to FITC, FAM, and Alexa Fluor 488. It can be useful for microscopy, fluorescence polarization, and selected binding assays.
LifeTein synthesized a peptide encompassing the ZnF region (a.a. 1–75) of human IRF2BPL with an N-terminal FITC tag linked through a 6-aminohexanoic acid (Ahx) spacer, C-terminal amidation, and >90% purity. This is a representative example of a long, fluorescently labeled peptide prepared for advanced cell and protein studies.
“FITC-Tet1, Alexa 594-N1, and Atto 643-N1 (95% purity) were acquired from LifeTein (NJ)...”

This case study shows a commonly used FRET design. The fluorescent dye MCA was incorporated at the N-terminus of a peptide substrate for stromelysin, a matrix metalloprotease. The quencher N-3-(2,4-dinitrophenyl)-L-2,3-diamino propionyl (DPA) was incorporated so that cleavage of the Gly-Leu bond separated donor and quencher.
MCA-Pro-Leu-Gly-Leu-DPA-Ala-Arg-NH2
MCA has peak excitation and emission at 328 nm and 393 nm. DPA has strong absorption at 363 nm with a shoulder at 410 nm, overlapping sufficiently with MCA fluorescence to allow quenching. Cleavage separates the two groups and increases fluorescence, allowing kinetic monitoring of matrix metalloprotease activity.
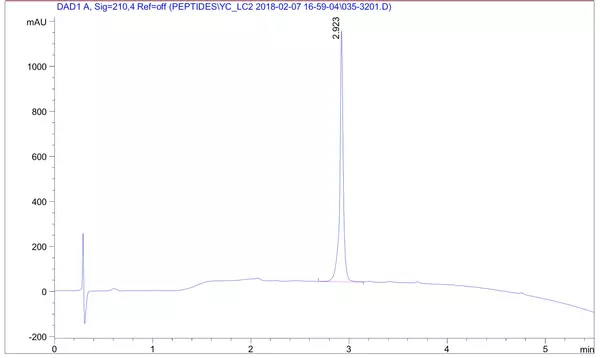
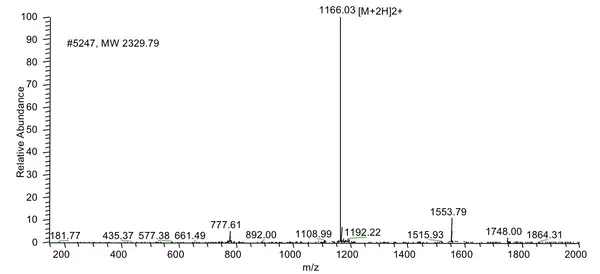
A client requested a very hydrophobic peptide 68 amino acids in length at 85% purity with FITC modification at the N-terminus. The peptide was synthesized successfully in 4 weeks.
HPLC Results:
MS Results:
LifeTein provides many fluorescent peptide labeling options, including FITC, FAM, AMCA, TAMRA, cyanine dyes, Alexa Fluor dyes, ATTO dyes, DyLight dyes, BODIPY, MCA, EDANS/Dabcyl, DNP, and other dye or quencher formats. Contact us if your preferred dye is not listed.
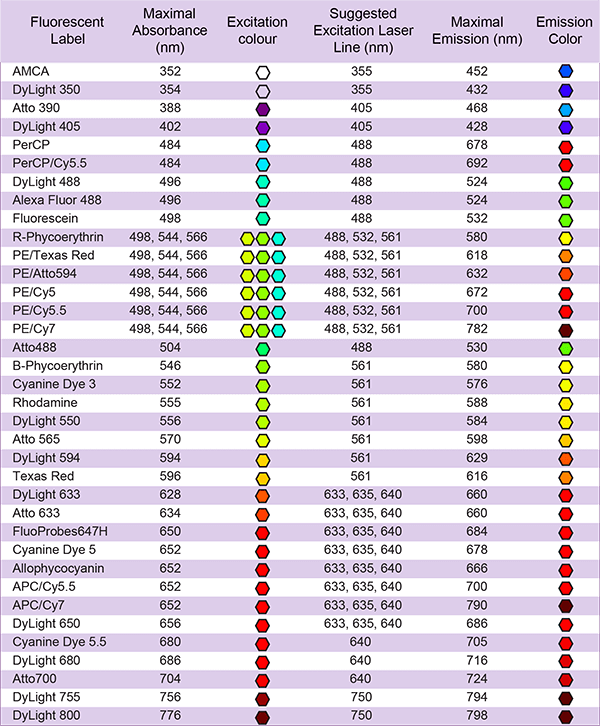
Individual dye pages are available in the fluorescent dye library: view fluorescent labeling options.
Please send us your peptide sequence, desired dye, labeling position, spacer preference, purity, quantity, and application. For FRET peptides, please include the donor, acceptor or quencher, cleavage site, and intended enzyme or assay.
If you already have a quote number, you can place your order online or send the signed quote form with your purchase order.