LifeTein provides peptide glycosylation services for research projects requiring defined glycopeptide structures, glycosylated amino acid building blocks, or custom carbohydrate-modified peptide constructs. These projects commonly involve O-linked or N-linked glycosylation motifs and are typically used in therapeutic peptide research, receptor studies, immunological studies, and structure–activity investigations.
Glycosylation is one of the most important natural peptide and protein modifications. In synthetic peptide chemistry, glycosylation is often used to explore how carbohydrate attachment affects peptide conformation, solubility, stability, biodistribution, receptor interaction, and membrane-facing behavior.
| Main formats | O-glycosylated peptides, N-glycosylated peptides, and selected custom glycopeptide formats |
| Common residues | Ser, Thr, and Asn glycosylation motifs, depending on target structure |
| Typical glycans | GlcNAc, GalNAc, mannose-related motifs, galactose-related motifs, and selected custom sugar-bearing amino acid building blocks |
| Typical synthesis route | Protected glycosylated amino acid building blocks incorporated during Fmoc solid-phase peptide synthesis |
| Project type | Primarily custom research-use synthesis rather than routine catalog production |
The exact effect depends strongly on the type of glycan, the attachment site, and the overall peptide sequence. Glycosylation can be beneficial, but it is not universally advantageous in every design.
N-glycosylation typically involves attachment to the amide nitrogen of Asn.
O-glycosylation typically involves attachment to the hydroxyl oxygen of Ser or Thr.
C-mannosylation is a more specialized form involving C–C linkage to Trp in selected biological systems.
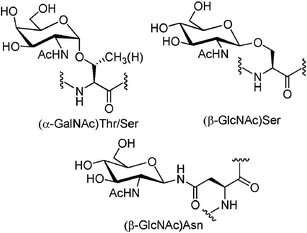
Illustration of O-linked serine/threonine glycosides and N-linked asparagine glycosides.
A common and practical strategy for synthetic glycopeptides is to use pre-formed, protected glycosylated amino acid building blocks that are compatible with Fmoc SPPS. This approach is well established for preparing defined glycopeptides and is generally more reliable than trying to glycosylate a completed peptide in a non-selective way.
For many projects, shorter or moderate-length glycopeptides are more practical than very long stepwise syntheses. As peptide length and complexity increase, incomplete coupling, steric hindrance, and overall yield can become more challenging.
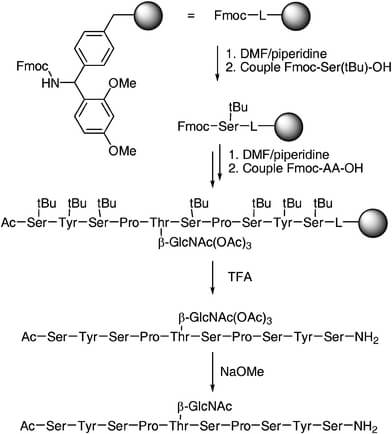
Example of a solid-phase approach for glycopeptide synthesis.
| Name | CAS | Formula |
|---|---|---|
| Fmoc-L-Ser((Ac)3-β-D-GlcNAc)-OH | 160067-63-0 | C32H36N2O13 |
| Fmoc-L-Thr((Ac)3-β-D-GlcNAc)-OH | 160168-40-1 | C33H38N2O13 |
| Fmoc-Asn(β-D-GlcNAc(Ac)3)-OH | 131287-39-3 | C33H37N3O13 |
| β-D-Glucose pentaacetate | 604-69-3 | C16H22O11 |
| Gluconic acid | 526-95-4 | C6H12O7 |
| 6-phosphogluconic acid | 921-62-0 | C6H13O10P |
| 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranosyl isothiocyanate | 14152-97-7 | C15H19NO9S |
| Name | CAS | Formula |
|---|---|---|
| Fmoc-L-Ser((Ac)3-β-D-GalNAc)-OH | 1676104-71-4 | C32H36N2O13 |
| Fmoc-L-Ser((Ac)3-α-D-GalNAc)-OH | 120173-57-1 | C32H36N2O13 |
| Fmoc-Thr(GalNAc(Ac)3-α-D)-OH | 116783-35-8 | C33H38N2O13 |
| Fmoc-L-Thr(β-D-GalNAc(Ac)3)-OH | 133575-43-6 | C33H38N2O13 |
| β-D-Galactose pentaacetate | 4163-60-4 | C16H22O11 |
| 1,2,3,4,6-Penta-O-acetyl-α-D-galactopyranose | 4163-59-1 | C16H22O11 |
| Name | CAS | Formula |
|---|---|---|
| Fmoc-Ser(Man(Ac)4-alpha-D)-OH | 118358-80-8 | C32H35NO14 |
| Fmoc-Thr(ManNAc)-OH | ||
| α-D-Mannose pentaacetate | 4163-65-9 | C16H22O11 |
| D-Mannose pentaacetate | 25941-03-1 | C16H22O11 |
| D-Mannopyranose tetraacetate | 140147-37-1 | C14H20O10 |
Best fit for this service
This service is best suited for researchers who already have a target glycopeptide concept, a defined glycosylation site, or a structure–activity question involving carbohydrate-modified peptides.
Related chemistry pages:
Need a quote?