Custom D-amino acid peptides, mirror-image peptides, retro-inverso peptides, and protease-resistant peptide analogs for stability, binding, and drug discovery studies.
LifeTein provides custom synthesis of peptides containing D-amino acids, fully D-enantiomeric mirror-image peptides, and retro-inverso peptide analogs. These designs are widely used to improve proteolytic stability, study peptide chirality, preserve epitope recognition, and develop more stable peptide-based research tools.
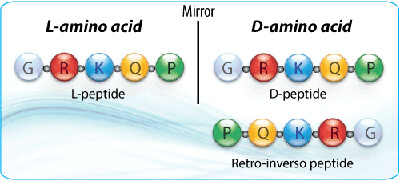
Most naturally occurring proteins and peptides are composed of L-amino acids. D-amino acids are the mirror-image stereoisomers of L-amino acids. Except for glycine, standard α-amino acids can exist in either L- or D-configuration.
Replacing selected L-amino acids with D-amino acids can significantly change peptide conformation, enzymatic stability, binding behavior, and biological activity. In many cases, D-amino acid substitution improves resistance to protease digestion while retaining key binding or recognition properties.

Mirror-image peptides are composed entirely of D-amino acids and represent the enantiomeric counterpart of the corresponding L-peptide. These peptides can show dramatically improved resistance to proteases because most biological proteases evolved to recognize L-amino acid peptide backbones.
Mirror-image peptides are useful in:
Retro-inverso peptides are made from D-amino acids arranged in the reverse order of the parent L-peptide sequence. This design is used to mimic the side-chain topology of the original peptide while reversing the peptide backbone direction.
Because the side chains may occupy a similar spatial orientation to the parent L-peptide, retro-inverso analogs can sometimes preserve biological recognition while gaining strong resistance to proteolytic degradation.
In some peptide designs, only the N-terminus, C-terminus, or flanking residues are replaced with D-amino acids. This approach can protect vulnerable terminal cleavage sites while preserving the central functional epitope or binding motif.
For example, studies with MUC2-derived peptides showed that adding D-amino acids to peptide flanking regions improved serum and lysosomal stability while maintaining antibody-binding properties.

D-amino acids can be used to tune peptide structure and stability. For example, D-Proline is often used to promote turn formation, while achiral residues such as Aib can influence helix formation and conformational control.
D-amino acid and retro-inverso designs are especially useful when the parent L-peptide is biologically active but unstable in serum or rapidly degraded by proteases.
D-amino acid substitutions can improve peptide resistance to proteolytic degradation. In one study, peptides containing D-amino acids at the N- and C-termini retained antibody-binding properties while becoming strongly resistant to degradation in diluted lysosomal and human serum preparations.
Lowercase letters are often used to indicate D-amino acids, while uppercase letters indicate L-amino acids. The addition of D-amino acids to both termini provided stronger protection than substitution at only one end.
A published study described a method to generate highly stable D-amino acid analogs of bioactive helical peptides using a mirror image of the Protein Data Bank. The authors designed D-peptide analogs based on mirror-image structures and evaluated critical binding residues.
LifeTein synthesized the peptides used in this study.
Please send your parent peptide sequence, desired D-amino acid substitutions, retro-inverso design, purity, quantity, and intended application. If you need help converting an L-peptide into a D-peptide or retro-inverso sequence, LifeTein can assist with design review.