
LifeTein provides high-quality amyloid peptides for Alzheimer’s disease and neurodegeneration research, including beta-amyloid sequences for aggregation studies, oligomer and fibril preparation, neurotoxicity assays, inhibitor screening, and structure–function investigations. Amyloid-β peptides remain among the most widely used research tools for understanding aggregation-prone peptide behavior and the molecular mechanisms associated with Alzheimer’s disease.
Among the available isoforms, Aβ (1-42) is especially important because its C-terminal extension increases hydrophobicity and strongly enhances aggregation propensity compared with Aβ (1-40). This property makes Aβ (1-42) a preferred sequence for studies of oligomer formation, fibrillization, neuronal toxicity, and amyloid plaque-related mechanisms.
| Main focus | Amyloid-β aggregation, oligomer toxicity, fibril formation, assay development, and Alzheimer’s disease-related mechanistic studies |
| Common formats | Aβ full-length peptides, short amyloid fragments, reverse-sequence controls, and sequence variants |
| Popular applications | Aggregation kinetics, Thioflavin T assays, neuronal toxicity models, compound screening, and structure–function analysis |
| Key product class | Beta-Amyloid (1-42), human |
| Category page | Amyloid peptide products |
Amyloid-β (1-42) is one of the most aggregation-prone forms derived from amyloid precursor protein (APP). Relative to shorter amyloid isoforms, Aβ42 more readily undergoes self-assembly into soluble oligomers, protofibrils, and mature fibrils. These aggregation states are associated with distinct biological properties and are widely studied in biochemical, cell-based, and translational Alzheimer’s research.
Published studies have shown that soluble oligomeric species are particularly relevant to early synaptic dysfunction, while more mature fibrillar structures are closely associated with amyloid plaque deposition. For this reason, Aβ (1-42) is commonly selected when researchers need a robust peptide model for both toxic oligomer formation and fibril-rich aggregation assays.
Aβ (1-42) undergoes a dynamic structural transition from relatively disordered peptide to assemblies enriched in cross-β structure. Depending on solvent history, concentration, ionic strength, and incubation conditions, the peptide can populate monomeric, oligomeric, protofibrillar, and mature fibrillar states. These forms are not interchangeable in experimental interpretation, and careful sample preparation is essential for reproducible results.
Compared with Aβ40, the extra C-terminal residues in Aβ42 promote stronger self-association and faster fibrillization. This difference is one reason Aβ42 is more frequently used in toxic oligomer studies and fibril-focused assay development.
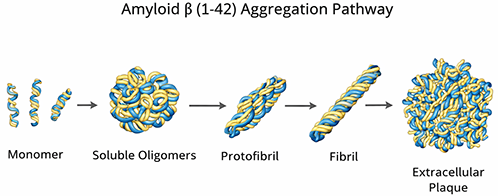
Monomeric Aβ (1-42) can assemble into soluble oligomers, protofibrils, mature fibrils, and ultimately plaque-like deposits.
Practical note
For amyloid work, the peptide sequence is only one part of the experiment. Solvent history, disaggregation treatment, incubation temperature, buffer choice, and storage conditions can all change the balance between monomeric, oligomeric, and fibrillar species.
Preparation of monomeric peptide
Preparation of oligomeric Aβ (ADDL-like conditions)
Preparation of fibrillar Aβ
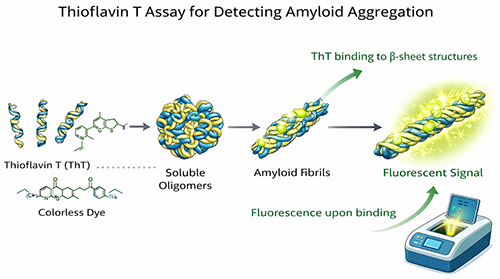
Thioflavin T (ThT) is one of the most widely used fluorescent dyes for monitoring amyloid fibril formation. Free ThT exhibits relatively weak fluorescence in solution, but the signal increases strongly when the dye binds to the ordered cross-β structure of amyloid fibrils. This makes the assay especially useful for monitoring fibril-rich samples, comparing aggregation kinetics, and screening compounds that modulate amyloid assembly.
For many amyloid fibril assays, fluorescence is typically measured using excitation near 450 nm and emission near 482 nm. ThT is particularly valuable for fibrillar Aβ samples, while early soluble oligomers are often better characterized with complementary techniques because ThT is primarily a fibril-focused probe.
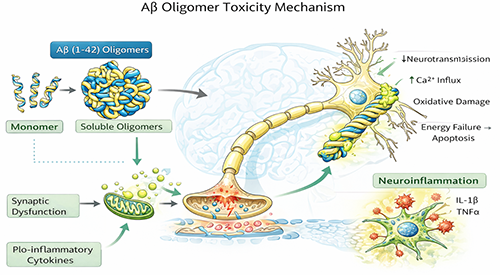
Soluble Aβ oligomers are widely studied because they are strongly associated with early neuronal dysfunction. Rather than acting only as passive precursors to plaques, these soluble assemblies can directly affect synaptic receptors, disrupt calcium homeostasis, alter membrane integrity, impair mitochondrial function, and promote inflammatory signaling in neural cells.
| Feature | Aβ (1-40) | Aβ (1-42) |
| Length | 40 amino acids | 42 amino acids |
| Aggregation propensity | Lower | Higher |
| Hydrophobicity | Moderate | Greater C-terminal hydrophobic character |
| Oligomer formation | Generally less aggregation-prone | Commonly used for stable toxic oligomer studies |
| Fibril formation | Slower kinetics | Faster fibrillization |
| Use in research | Isoform comparison and aggregation studies | Alzheimer’s disease-focused toxicity and aggregation studies |
LifeTein offers multiple amyloid-related products that can be used together for aggregation, control, and structure–function studies.
| Product | Main Use | Link |
| Beta-Amyloid (1-42), human | Aggregation, oligomer toxicity, fibril formation, Alzheimer’s disease model assays | View product |
| Beta-Amyloid (1-40), Ultra Pure, TFA | Isoform comparison, aggregation kinetics, Aβ40 vs Aβ42 studies | View product |
| Amyloid category | Browse full amyloid peptide offering | View category |
Because amyloid peptides are highly aggregation-prone, reproducibility depends strongly on preparation and storage. For practical handling guidance, see: