PeptideSynTM is LifeTein’s peptide synthesis technology platform for efficient AI-assisted peptide design and manufacturing, rapid microwave-assisted synthesis, and flexible process design for difficult peptide projects. It supports long peptides, modified peptides, and sequence-specific synthesis strategies while helping improve turnaround time, synthesis efficiency, and overall product quality.
| Core platform | AI-assisted peptide design and manufacturing, Microwave-assisted solid-phase peptide synthesis combined with flexible coupling chemistry and practical workflow design |
| Applications | Long peptides, hydrophobic peptides, modified peptides, peptide-protein conjugates, and challenging custom sequences |
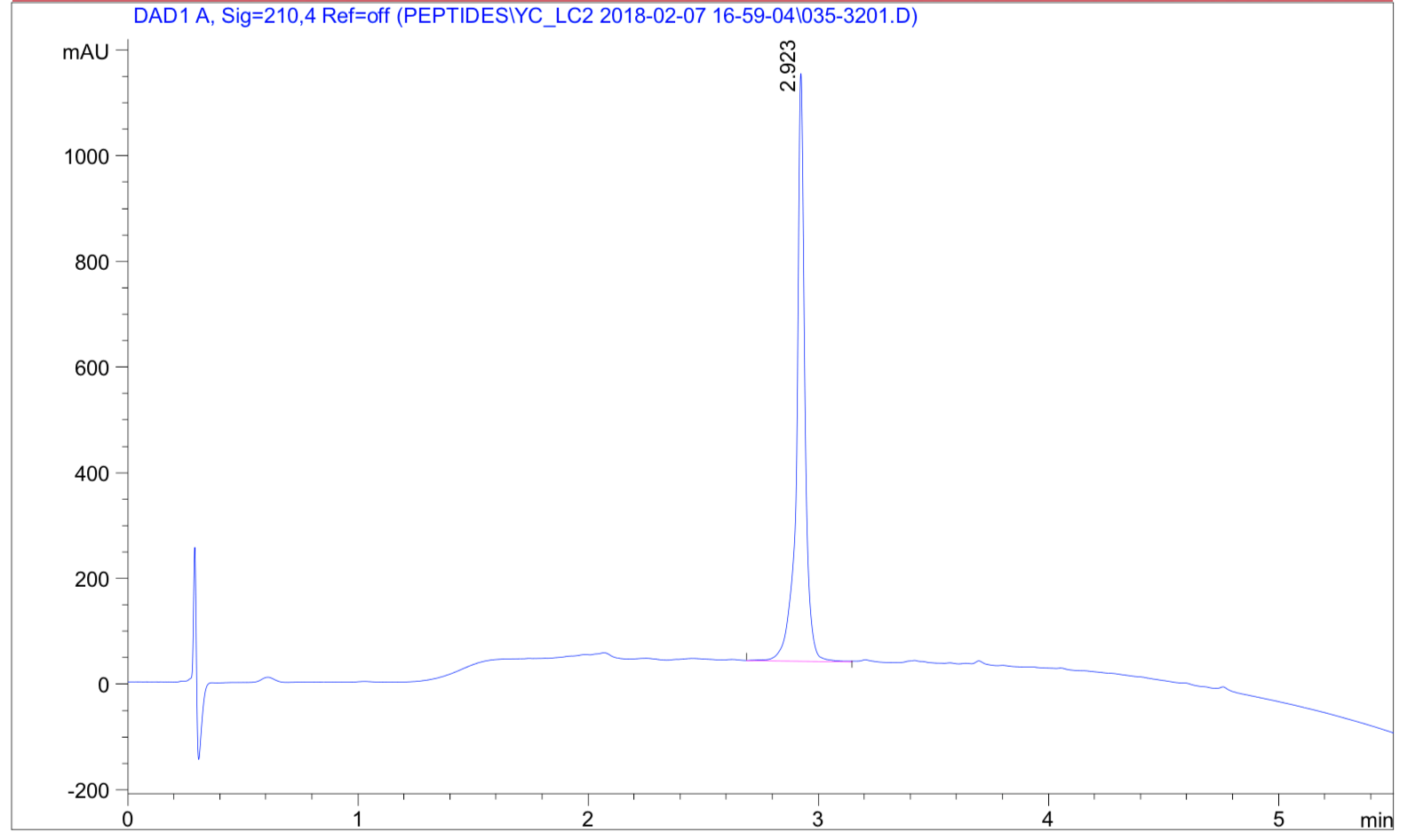
| Analytical support | Bio-inert HPLC system with improved recovery, peak consistency, and reproducibility under demanding analytical conditions |
| Coupling flexibility | Compatible with multiple coupling reagents including HBTU, HATU, HCTU, ByBroP, BOP, PyBOP, and TBTU |
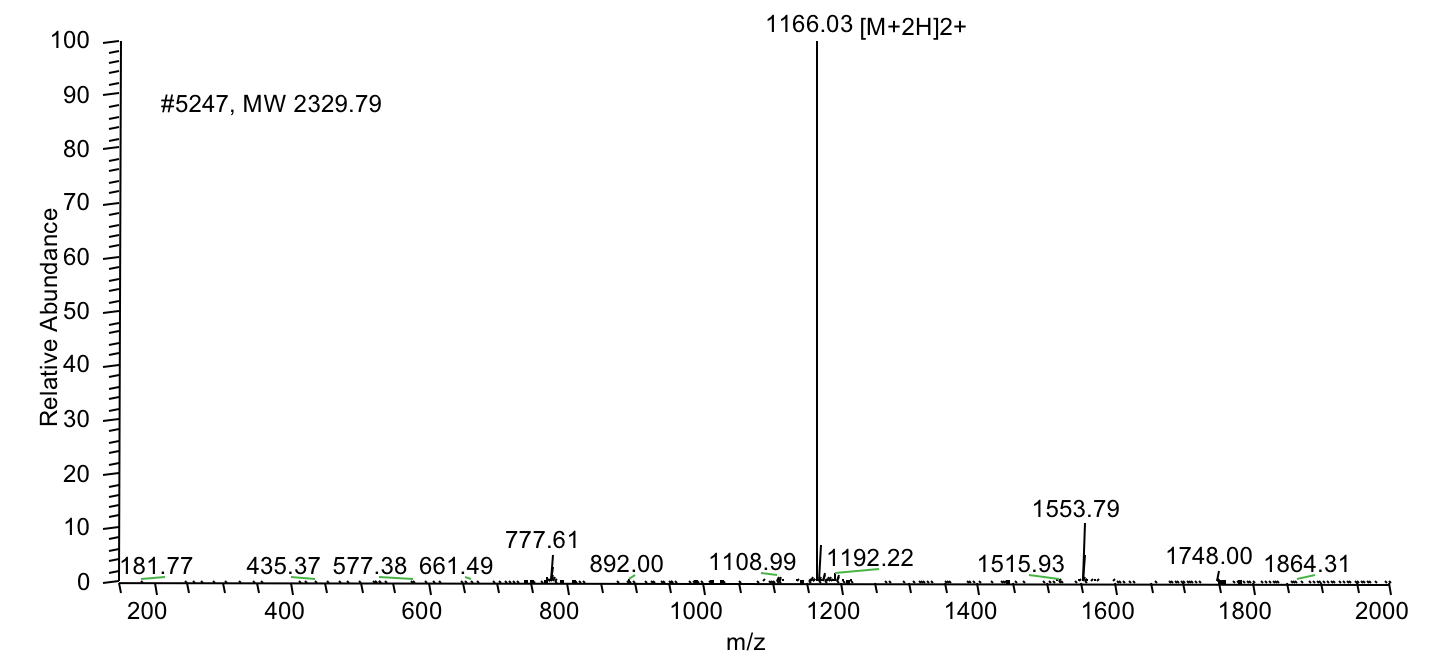
| Demonstrated capability | Peptides exceeding 100 amino acids, including 76aa, 97aa, 133aa, and 169aa peptide projects |
Microwave synthesis is a practical method used by LifeTein to accelerate peptide synthesis. This technique rapidly heats solvents or amino acids in solution through dipolar rotation or ionic conduction, enabling efficient and controlled heating during peptide assembly. For many peptide projects, this helps reduce synthesis time while maintaining practical process control.
LifeTein’s implementation of a bio-inert HPLC system provides additional advantages for peptide purification and analysis, including improved recovery, better sensitivity, reliable peak consistency, robustness under harsh buffer conditions, and support for multiple chromatographic methods. These capabilities help us deliver higher-quality peptides, peptide-protein conjugates, labeled molecules, and other modified biomolecules with stringent QC and reproducible methodologies.
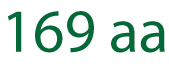 |
LifeTein has successfully used proprietary technology to produce peptides exceeding 100 amino acids with AI-assisted peptide design and manufacturing. Our notable achievements include a 76aa BfUbb peptide, a 97aa peptide mB Box-97syn, a 133aa SHT peptide, and a 169aa long peptide. |
PeptideSynTM is also suitable for advanced modified peptide formats. For example, LifeTein provides Peptide Oligonucleotide Conjugates and other specialized modified peptides that require careful coordination of synthesis, coupling chemistry, and downstream purification.
Conventional solid-phase peptide synthesis (SPPS) often involves long coupling cycles, extended deprotection periods, repeated washing steps, and Kaiser tests for acylation confirmation. These workflows can become especially challenging for complex peptides such as beta-amyloid, aggregation-prone sequences, or large enzyme-related peptides, where on-resin aggregation may reduce synthesis performance.
LifeTein addresses these challenges with a platform designed for rapid assembly of peptide fragments using suitable coupling chemistry, native chemical ligation where appropriate, and microwave-assisted heating. PeptideSynTM provides a practical synthesis platform for long peptides and advanced research peptides, including sequences containing D-amino acids or pseudoproline dipeptides.
The flexibility of PeptideSynTM allows selection among efficient coupling reagents such as HBTU, HATU, HCTU, ByBroP, BOP, PyBOP, and TBTU, supporting fast and adaptable SPPS workflows.
Need help with a difficult peptide sequence?
Send us your peptide sequence, purity target, and any modification requirements. We can review whether PeptideSynTM is a good fit for your project.
In one study using PeptideSynTM technology, the synthesis of Amylin (1-37), human {KCNTATCATQRLANFLVHSSNNFGAILSSTNVGSNTY (Disulfide bridge: 2-7)}, and human beta-amyloid (1-42) (H-DAEFRHDSGYEVHHQKLVFFAEDVGSNKGAIIGLMVGGVVIA-OH) using HCTU as an activator required only about 15 hours of synthesis time, while producing peptides with purity comparable to peptides made using much longer traditional methods.
Coupling yields for the peptide segment were approximately 90–95%, illustrating the efficiency and practical utility of PeptideSynTM technology for demanding peptide targets.
By combining suitable coupling reagents, appropriate deprotection strategies, and the correct resin selection, LifeTein uses PeptideSynTM to reduce overall peptide synthesis time and improve peptide quality for many project types.
In another project, a client requested synthesis of a highly hydrophobic 68-amino-acid peptide at 85% purity with a fluorescent FITC modification at the N-terminus. LifeTein successfully completed this challenging synthesis within 2 weeks, demonstrating the platform’s ability to support difficult sequence properties together with dye modification requirements.