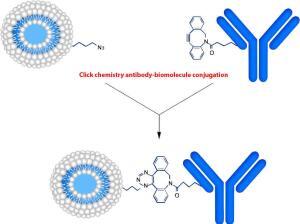
We describe a simple method for preparing antibody-peptide, antibody-oligonucleotide, or antibody-compound conjugates and discuss its applications in drug delivery and new drug design. Conjugation is based on alkyne-azide cycloaddition. This Cu-free click reaction starts from the dibenzocyclooctyne (DBCO) moiety-activated antibodies and subsequently linked covalently with an azide-modified peptide, oligonucleotide or compounds. The reaction is performed under physiological conditions and has no adverse effects on antibodies or proteins. This can also be used as the click chemistry fluorescence labeling and the click chemistry in peptide-based drug design.
However, the copper-catalyzed alkyne-azide cycloaddition (CuAAC) is unsuitable for functional biomolecule applications because copper ions can cause protein denaturation.
Measuring the protein levels directly is challenging. However, the signals can be amplified by immuno-PCR using oligonucleotide-attached antibodies to detect protein indirectly.

Antibody-Conjugate
Preparing Antibody-Peptide, Antibody Oligonucleotide or Antibody-Compound Conjugates
1. Conjugation of DBCO to the Antibody. The DBCO-PEG5-NHS was used to react with the NH2 groups on the antibody. The inclusion of a PEG5 linker improves the water solubility of the hydrophobic DBCO, introduces a spacer, and increases flexibility between the antibody molecule and the peptide/oligonucleotide or compounds. This will alleviate the antibody’s steric effect on the enzymatic reactions.
2. Prepare the azido-peptide or azido-oligonucleotide. LifeTein provides click chemistry modified peptide synthesis: N-terminal azide-peptide/oligo or C-terminal peptide/oligo-azide.
3. Covalent attachment of the peptide/oligonucleotide to the antibody. The reaction between DBCO and azide is slow compared to the CuAAC reaction. The 16–18 h reaction time in PBS at 4 °C is ideal for increasing the final product yield. The DBCO-antibody in the intermediate reaction is stable.
https://pubs.acs.org/doi/full/10.1021/acs.bioconjchem.5b00613