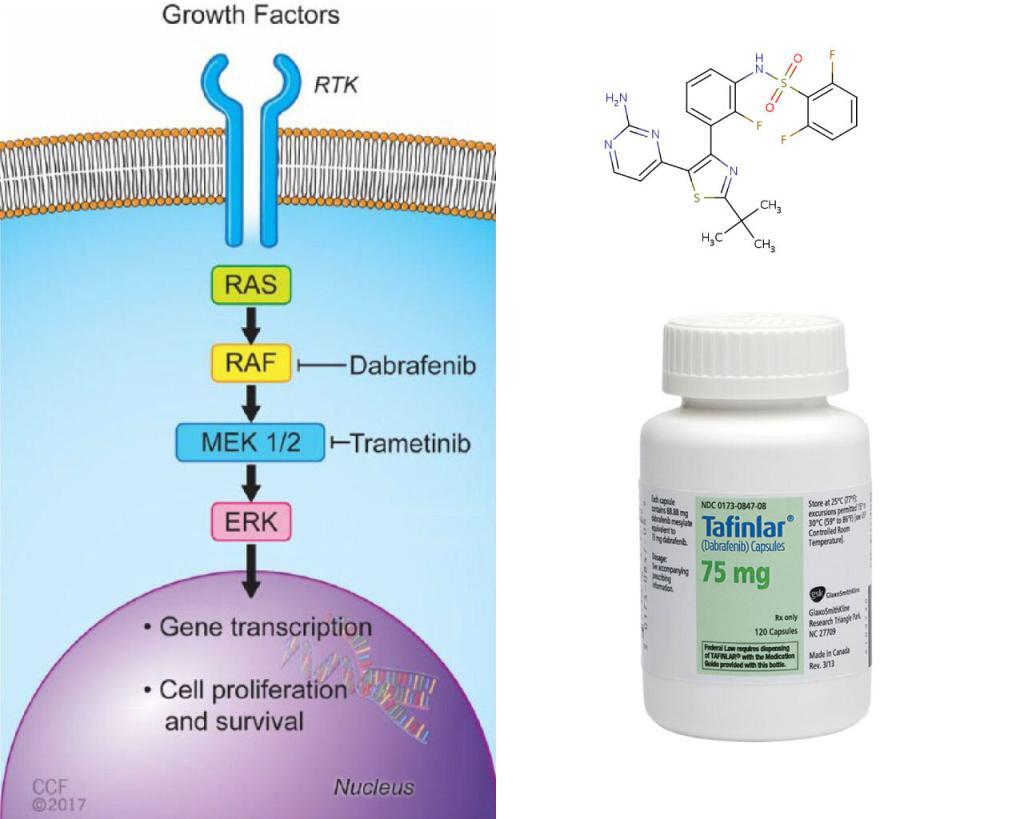
BRAF is an RAF kinase. It is a core component of the RAS/RAF/MEK/ERK signaling cascade, known as mitogen-activated protein kinase (MAPK) pathway. It is one of the major effectors of oncogene RAS, and is often mutated in human cancer cells.
LifeTein’s Braftide & Cancer Therapy
Two FDA approved drugs, Dabrafenib, and vemurafenib, effectively inhibit the most common BRAF variant V600E, a monomeric BRAF. But, the non-V600E BRAF mutations are intrinsically resistant to these drugs. These drugs may also paradoxically stimulate the pathway when the tumor cells contain wild-type BRAF and oncogenic RAS, causing secondary malignancies.
The researchers tried to tackle the dimeric BRAF. The dimeric BRAF, such as the wild type and G469A, a most prevalent non-V600E variant in lung cancer cells, hinges on dimer interface (DIF), a 20aa span near the tail end of the alpha-C helix of BRAF. The researchers designed Braftide using computational modeling, aiming to block the dimerization. They tested the functionality in vitro, in HEK263 cells and colon cancer cell lines.
LifeTein synthesized Braftide (TRHVNILLFM), Null-Braftide (THHVNILLFM), Cy3-Braftide (TRHVNILLFM-Cy3), TAT-Braftide (GRKKRRQRRRPQ-PEG-TRHVNILLFM), and TAT (GRKKRRQRRRPQ). We reviewed here some of the assays that helped support Braftide as an allosteric inhibitor of BRAF dimer and down-regulator of MAPK signaling pathway for cancer therapy.
1) Cell-free in vitro assay: dose-response curve. First of all, the researchers show that Braftide has a sub-micromolar IC50 for dimeric BRAF. Full-length dimeric BRAF-WT and BRAF-G469A (from HEK293F cells) were used for dose-response curves, and the BRAF activity was probed by pMEK production.
2) Cell-free in vitro assay: Saturation binding assay. The researchers used Cy3-labeled Braftide (Cy3-Braftide) to characterize (KD) the binding of Braftide with dimeric BRAF-WT using fluorescence quantification.
3) Cell-free in vitro assay: Immunoprecipitation (IP). The purpose of IP was to show Braftide disrupted the BRAF dimerization. Braftide was added to HEK293 cell lysate coexpressing V5- and FLAG-tagged BRAF-WT. FLAG-tagged BRAF was pulled down by FLAG antibody-conjugated resin, which was further probed for V5-tagged BRAF. Braftide indeed reduced homodimer BRAF.
4) Delivery of Braftide into HEK cell for BRAF inhibition. Braftide was tagged with cell-penetrating peptide TAT. TAT-Braftide (and its negative control TAT alone) was used to treat HEK293 cells transiently transfected with BRAF-WT and BRAF-G469A. Four hours of treatment resulted in reductions of BRAF, pMEK, MEK (i.e. the MAPK pathway), which were analyzed with respective antibodies by immunoblotting.
5) Delivery of Braftide into cancer cells for BRAF inhibition and cell proliferation inhibition. Two colon cancer cell lines (KRAS-G13D-colon carcinoma) were treated with cell-penetrating TAT-Braftide and assayed for the inhibition of BRAF activity, down-regulation of MAPK signaling, and cell proliferation. All were shown positive, while the negative control TAT alone were negative.
https://pubs.acs.org/doi/abs/10.1021/acschembio.9b00191
Our Services:
Custom Peptide Synthesis Services
Other Posts:
Smaller Ions Stabilize β-sheets
